There are a number of methods for generating graphene and chemically modified graphene from graphite and derivatives of graphite, each with different advantages and disadvantages. A new approach is to use colloidal suspensions to produce new materials composed of graphene and chemically modified graphene. This approach is both versatile and scalable, and is adaptable to a wide variety of applications.
Graphene has been made by four different methods. The first was chemical vapour deposition (CVD) and epitaxial growth, such as the decomposition of ethylene on nickel surfaces. These early efforts (which started in 1970) were followed by a large body of work by the surface-science community on ‘monolayer graphite’. The second was the micromechanical exfoliation of graphite. This approach, which is also known as the ‘Scotch tape’ or peel-off method, followed on from earlier work on micromechanical exfoliation from patterned graphite. The third method was epitaxial growth on electrically insulating surfaces such as SiC and the fourth was the creation of colloidal suspensions.
Properties of Graphene
The remarkable properties of graphene reported so far include high values of its Young’s modulus (approx 1,100 GPa), fracture strength (125 GPa), thermal conductivity (approx 5,000 W m-1K-1), mobility of charge carriers (200,000 cm2 V-1 s-1) and specific surface area (calculated value, 2,630 m2 g-1), plus fascinating transport phenomena such as the quantum Hall effect. Graphene and chemically modified graphene (CMG) are promising candidates as components in applications such as energy-storage materials, ‘paper-like’ materials, polymer composites, liquid crystal devices and mechanical resonators.
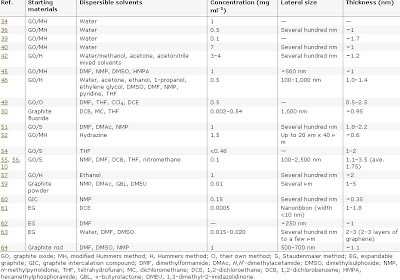
Comparison of a set of chemical approaches to produce colloidal suspensions of CMG sheets

Challenges and Perspectives
Colloidal suspensions are of great importance in the preparation of many types of materials, and the suspensions of chemically modified graphene (CMG)s hold great promise in this regard. Looming issues in terms of wide-scale applicability include scalability (yield, quantity, cost, etc.), the safety of the solvents used and the removal from the product material (if necessary) of residual solvents or stabilizers used in the colloid. It is also worth emphasizing that although colloidal suspensions are normally only regarded as stable if they persist for very long periods of time, dispersions of CMGs might remain stable long enough to be processed into something else.
Another critical issue is related to our understanding of the chemical structure(s) of CMG sheets and their reaction mechanisms. The better our knowledge of the chemistry of these materials, the better the graphene-based composites, thin films, paper-like materials and so on that we can make. For example, the prospects for sensors based on CMG will hinge on our ability to chemically tune the CMG for each sensing modality.
So far the graphenes derived by the reduction of graphene oxide have contained a significant amount of oxygen and, possibly, significant numbers of defects. Thermal annealing of reduced graphene oxide sheets has produced enhanced results and finding routes for complete restoration of the sp2 carbon network of pristine graphene is of interest. (The graphenes produced from graphite intercalation compounds or expandable graphite may have fewer defects, although they are also produced in lower yields and are less amenable to functionalization than graphenes derived from graphene oxide.)
Finally, we mention the development of new reaction routes and starting materials as an alternative. The worldwide supply of natural graphite has been estimated at 800,000,000 tonnes. If graphene or very thin platelets of multilayer graphene could be produced on a large scale by CVD from various precursors, new routes for creating colloidal suspensions might also be found, and the supply of graphene/few-layer graphene might be enormously increased.

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.