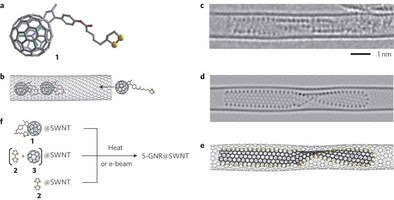
Carbon nanotubes serve as containers and nanoreactors for molecules.
Researchers have demonstrated that carbon nanotubes can be used as nanoscale chemical reactors and chemical reactions involving carbon and sulphur atoms held within a nanotube lead to the formation of atomically thin strips of carbon, known as graphene nanoribbon, decorated with sulphur atoms around the edge.
The ability to tune the properties of graphene nanoribbons (GNRs) through modification of the nanoribbon’s width and edge structure widens the potential applications of graphene in electronic devices. Although assembly of GNRs has been recently possible, current methods suffer from limited control of their atomic structure or require the careful organization of precursors on atomically flat surfaces under ultra-high vacuum conditions. Here we demonstrate that a GNR can self-assemble from a random mixture of molecular precursors within a single-walled carbon nanotube, which ensures propagation of the nanoribbon in one dimension and determines its width. The sulphur-terminated dangling bonds of the GNR make these otherwise unstable nanoribbons thermodynamically viable over other forms of carbon. Electron microscopy reveals elliptical distortion of the nanotube, as well as helical twist and screw-like motion of the nanoribbon. These effects suggest novel ways of controlling the properties of these nanomaterials, such as the electronic band gap and the concentration of charge carriers.
Dr Khlobystov said: “Graphene nanoribbons possess a wealth of interesting physical properties making them more suitable for applications in electronic and spintronic devices than the parent material graphene — the discovery of which attracted the Nobel Prize for Physics last year for University of Manchester scientists Professors Andre Geim and Konstantin Novoselov.
“Nanoribbons are very difficult to make but the Nottingham team’s strategy of confining chemical reactions at the nanoscale sparks spontaneous formation of these remarkable structures. The team has also discovered that nanoribbons — far from being simple flat and linear structures — possess an unprecedented helical twist that changes over time, giving scientists a way of controlling physical properties of the nanoribbon, such as electrical conductivity.”
Devices based on nanoribbons could potentially be used as nano-switches, nano-actuators and nano-transistors integrated in computers or data storage devices.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.