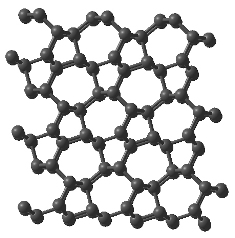
Researchers used a novel computational method to demonstrate that the properties of what had previously been thought to be only a hypothetical structure of a superhard form of carbon called “M-carbon” – constructed by Oganov in 2006 – matched perfectly the experimental data on “superhard graphite.” M-carbon is almost as hard as diamond.
Another result of this study is a set of detailed mechanisms of formation of several potential carbon allotropes. These could be used to engineer ways of their synthesis for potential technological applications.
“We don’t know yet which applications M-carbon will find, but most forms of carbon did manage to find revolutionary applications, and this amazing material might do so as well,” said Oganov.
The story of yet another form of carbon started in 1963, when Aust and Drickamer compressed graphite at room temperature. High-temperature compression of graphite is known to produce diamond, but at room temperature an unknown form of carbon was produced. This new form, like diamond, was transparent and superhard – but its other properties were inconsistent with diamond or other known forms of carbon.
“The experiment itself is simple and striking: you compress black ultrasoft graphite, and then it suddenly turns into a colorless, transparent, superhard and mysterious new form of carbon – ‘superhard graphite,’” said Oganov. “The experiment was repeated several times since, and the result was the same, but no convincing structural model was produced, due to the low resolution of experimental data.”
The new work confirms that experiments produced M-carbon, which has a known structure.
M-carbon is expected to be almost as hard as diamond.
If Superman crushes Graphite he gets M-Carbon unless he adds a lot of heat
According to Oganov, the reason why diamond is not formed on cold compression of graphite is that the reconstruction needed to transform graphite into diamond is too large and is associated with too great an energy barrier, which can be overcome only at high temperatures, when atoms can jump far. At low temperatures, graphite chooses instead a transformation associated with the lowest activation barrier.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.