Continuing the Alzheimer’s and Neurodegenerative
Diseases track in the Rejuvenation Biotechnology Conference, Jane Lebkowski, President of R&D, Asterias Biotherapeutics Inc.
Diseases track in the Rejuvenation Biotechnology Conference, Jane Lebkowski, President of R&D, Asterias Biotherapeutics Inc.
discussed her group’s work on the development of pluripotent stem cell derived oligodendrocyte progenitor cells for the treatment of spinal cord injury. There are a myriad of after effects of spinal cord injury, including loss of limb function, impaired cardiovascular function, loss of control over bowel and bladder movements, neurogenic pain, affected sexual function, spasticity, debilitating sensations, among others. This costs the USA an average of $12k/patient/year, in mostly younger men, but we are seeing these types of injuries in older men as well. Of course women of all ages are affected, but due to the popularity of action sports, we still see a higher male demographic.
There are many considerations in developing stem cell-based therapeutic and oligodendrocyte
progenitor cells may serve as a good platform for restoring some function and improve the life quality of those suffering from the effects of spinal cord injury.
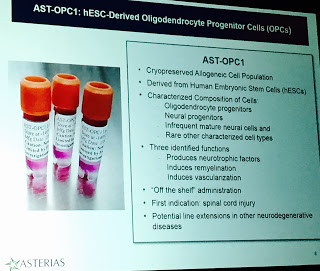
AST-OPC1 are hESC-derived oligodendrocyte progenitor cells that have been characterized and could be administered ‘off the shelf.’ Studies in animal models use transplanted cells to evaluate their effects. They found that 7-10d post injury, there was an increase in limb coordination, decrease in tail drag, better weight bearing and trunk stability.
Months after, there is also improvement in locomotor activity. The transplant also increased surviving cells and myelinated fibers that traverse injury site at the cervical location. In 2010 after performing almost 30 animal studies, they got FDA clearance to move into clinicals. The thoracic SCI trial was launched, but these patients will be followed over the next fourteen years, with multiple MRIs for tumor growth and degeration. So far there were no immune responses, stable engraftment, and no changes in neurological function. But also, no efficacy anticipated due to low dose. So they’ve moved into another set of trials looking at dose-response.
A question from the audience was: Why are these cells dose dependent? Lebkowski explained that they are not proliferative but they do migrate -so there is probably a dose effect to get sufficient cells migrating throughout injury site.
Strides in the spinal cord injury field, but we are still far from seeing any dramatic rejuvenation of the spine in this population.