A team at MIT has come up with an innovative desalination approach that, unlike most traditional desalination systems, does not separate ions or water molecules with filters, which can become clogged, or boiling, which consumes great amounts of energy.
Instead, the system uses an electrically driven shockwave within a stream of flowing water, which pushes salty water to one side of the flow and fresh water to the other, allowing easy separation of the two streams.
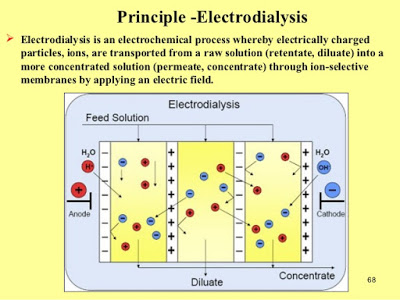
Membranes in traditional desalination systems, such as those that use reverse osmosis or electrodialysis, are “selective barriers,” Bazant explains: They allow molecules of water to pass through, but block the larger sodium and chlorine atoms of salt. Compared to conventional electrodialysis, “This process looks similar, but it’s fundamentally different,” he says.
In the new process, called shock electrodialysis, water flows through a porous material —in this case, made of tiny glass particles, called a frit — with membranes or electrodes sandwiching the porous material on each side. When an electric current flows through the system, the salty water divides into regions where the salt concentration is either depleted or enriched. When that current is increased to a certain point, it generates a shockwave between these two zones, sharply dividing the streams and allowing the fresh and salty regions to be separated by a simple physical barrier at the center of the flow.
“It generates a very strong gradient,” Bazant says.
Even though the system can use membranes on each side of the porous material, Bazant explains, the water flows across those membranes, not through them. That means they are not as vulnerable to fouling — a buildup of filtered material — or to degradation due to water pressure, as happens with conventional membrane-based desalination, including conventional electrodialysis. “The salt doesn’t have to push through something,” Bazant says. The charged salt particles, or ions, “just move to one side,” he says.
The underlying phenomenon of generating a shockwave of salt concentration was discovered a few years ago by the group of Juan Santiago at Stanford University. But that finding, which involved experiments with a tiny microfluidic device and no flowing water, was not used to remove salt from the water, says Bazant, who is currently on sabbatical at Stanford.
The new system, by contrast, is a continuous process, using water flowing through cheap porous media, that should be relatively easy to scale up for desalination or water purification. “The breakthrough here is the engineering [of a practical system],” Bazant says.
One possible application would be in cleaning the vast amounts of wastewater generated by hydraulic fracturing, or fracking. This contaminated water tends to be salty, sometimes with trace amounts of toxic ions, so finding a practical and inexpensive way of cleaning it would be highly desirable. This system not only removes salt, but also a wide variety of other contaminants — and because of the electrical current passing through, it may also sterilize the stream. “The electric fields are pretty high, so we may be able to kill the bacteria,” Schlumpberger says.
Abstract
Rising global demand for potable water is driving innovation in water treatment methods. Shock electrodialysis is a recently proposed technique that exploits deionization shock waves in porous media to purify water. In this letter, we present the first continuous and scalable shock electrodialysis system and demonstrate the separation of sodium, chloride, and other ions from a feed stream. Our prototype continuously removes over 99% (and up to 99.99%) of salt from diverse electrolytes over a range of concentrations (1 mM, 10 mM, and 100 mM). The desalination data collapses with dimensionless current, scaled to charge advection in the feed stream. Enhanced water recovery with increasing current (up to 79%) is a fortuitous discovery, which we attribute to electro-osmotic pumping. The results suggest the feasibility of using shock electrodialysis for practical water purification applications.

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.