Diamonds are formed from the same carbon atoms that also make up the soft graphite found in the centre of pencils. The difference between these two forms of carbon is the arrangement of the atoms The search for superhard materials begins by attempting to replicate the structure of diamond, but there are only a few elements able to bind together in this way.
One such material is boron nitride. Like carbon, this synthetic material comes in several different forms, but it is possible to replicate the structure of diamond by replacing the carbon atoms with nitrogen and boron atoms. First created in 1957 and known as cubic boron nitride, it was initially reported to be hard enough to scratch diamond – hopes that quickly dulled as later tests showed that it is less than half as hard as its carbon-based counterpart.
It was not until 2001 that a diamond-like boron carbon nitride was reported to have been produced by researchers at the National Academy of Sciences of Ukraine in Kiev with colleagues in France and Germany. But they found while the new material was harder than crystals of cubic boron nitride it was still fell short of diamond.
Seven years ago, Changfeng Chen, a physicist at the University of Nevada, and colleagues at Shanghai Jiao Tong University in China, thought they had hit on something that might topple diamond from its pedestal. They calculated that a bizarre hexagonal form of boron nitride, known as wurtzite boron nitride would be able to resist 18% more stress than diamond.
Wurtzite boron nitride becomes nearly 80% stronger under higher pressures. The snag is that wurtzite boron nitride is rather dangerous to create – it only occurs naturally in the extreme heat and pressure of volcanic eruptions and has to be created synthetically in explosions that mimic these conditions, meaning it is notoriously difficult to obtain in sufficient quantities and it has yet to be tested. Similar problems have limited the potential study of a related substance, known as lonsdaleite, that should be able to withstand up to 58% more stress than standard diamond crystals.
In 2015, Jagdish Narayan, and his colleagues at North Carolina State University revealed they had melted a non-crystalline form of carbon known as glassy-carbon with a rapid laser pulse, heating it to 3,700C (6690F) before rapidly cooling it. This cooling, or quenching, step led to the name Q-carbon. What they had produced was a strange, but exceptionally strong amorphous form of carbon. Unlike other forms of carbon it is magnetic and glows when exposed to light.
The structure of the material itself was mainly constructed from diamond-type bonding but also had about 10 to 15% graphite-type bonding. Tests by the research team suggest Q-carbon could be at least 60% harder than diamond, but this has yet to be definitively confirmed.
When crushing a sample of Q-carbon between two sharpened diamond points, this creates a problem. “The diamond tips deformed during hardness measurements of Q-carbon,” said Narayan.
Nanocrystalline diamond balls made of lots of tiny diamond crystals bound by graphene are over 4 times stronger than regular diamond crystal
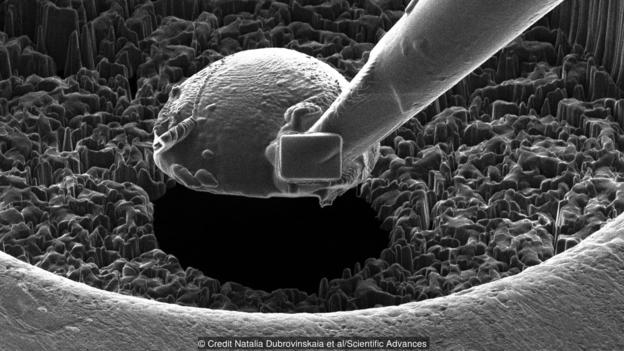
It is here that Dubrovinskaia’s superhard anvils enter the picture. Her new material is a unique form of carbon known as nanocrystalline diamond balls, and rather than being made from a single crystal lattice of carbon atoms like the gemstones we prize for engagement rings, it is made up of lots of tiny individual crystals – each 11,000 times smaller than the width of a human hair – that are bound to each other by a layer of graphene.
Whereas a diamond crystal will begin to give way at pressures of up to 120 Giga Pascals (GPa), the new material, however, can withstand at least 460 GPa. It can even survive when pressed together to generate pressures of up to 1,000 GPa. That makes these tiny spheres harder than any other known substance on the planet.
Nanocrystalline diamond balls such as this are thought to be the hardest materials known to humankind (Credit Natalia Dubrovinskaia et al/Scientific Advances)
Higher pressures to get metallic hydrogen
Squeeze anything hard enough and it will become a metal. There is a global race to advance high pressure science to create metallic hydrogen. Metallic hydrogen might be superconducting and it might continue to be metallic after the pressure is removed.
If solid metallic hydrogen turns out to be a room-temperature superconductor, it would have to be crushed to work, making it impractical for many applications. But if hydrogen could hold its metallic form after the pressure is released, as some researchers have suggested, “it would be revolutionary,” says physicist Isaac Silvera, who leads the metallic hydrogen hunt at Harvard University. Such a material could be used in electrical wires to reduce loss of energy and decrease the world’s power consumption. And it might lead to efficient, magnetically levitated trains and technological advances in nuclear fusion, supercomputing and more.
While one group of would-be metallurgists is searching for solid metal, other investigators seek the scientifically intriguing liquid hydrogen metal.
SOURCES – BBC News, Science News

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.