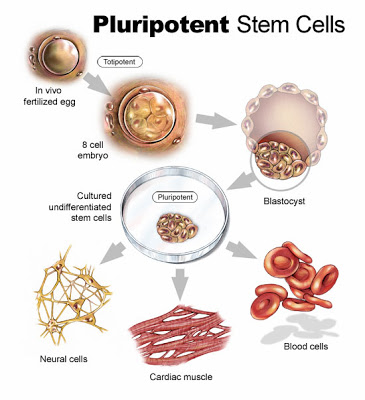
1. Researchers have created healthy stem cells from adult cells–no embryo required and no side effects.
Until now, however, creating iPS [induced pluripotent stem] cells without integrated viruses had been a major hurdle for stem-cell researchers. Although Hochedlinger has overcome that hurdle, he says there is still some distance to travel. While retroviral techniques allow scientists to turn about one in every 1,000 skin cells into an iPS cell, the adenovirus is far less efficient: only one in every 10,000 to 100,000 fetal liver cells can be converted.
Once there are more iPS cells they can be compared to embryonic stem cells to confirm that they are identical.
2. Singapore’s Institute of Bioengineering and Nanotechnology Announces: Reversible 3D Cell Culture Gel Invented by IBN Published in Nature Nanotechnology. This should help with the study and use of stem cells and increase the flexibility of working with cells.
A thixotropic nanocomposite gel for three-dimensional cell culture
Y. Shona Pek, Andrew C. A. Wan, Asha Shekaran, Lang Zhuo & Jackie Y. Ying
Thixotropic materials, which become less viscous under stress and return to their original state when stress is removed, have been used to deliver gel–cell constructs and therapeutic agents. Here we show that a polymer–silica nanocomposite thixotropic gel can be used as a three-dimensional cell culture material. The gel liquefies when vortexed—allowing cells and biological components to be added—and resolidifies to trap the components when the shear force from spinning is removed. Good permeability of nutrients and gases through the gel allows various cell types to proliferate and be viable for up to three weeks. Human mesenchymal stem cells cultured in stiffer gels developed bone-like behaviour, showing that the rheological properties of the gel can control cell differentiation. No enzymatic, chemical, or photo-crosslinking, changes in ionic strength or temperature are required to form or liquefy the gel, offering a way to sub-culture cells without using trypsin—a protease commonly used in traditional cell culture techniques.
The research teams in Denver and Rochester consider the dramatically dissimilar outcomes between the different astrocyte transplants a development that can change the way stem cell technologies are used to repair spinal cord injuries. To that end, the researchers are in the process of developing a safe, efficient and cost-effective way to use this approach to better define the optimal human astrocytes with an eye toward use for clinical trials.
Researchers transplanted 200,000 embryonic stem cells into the wall of the left ventricle of the knockout mice. After one month the treatment improved heart performance, synchronized electrical impulses and stopped heart deterioration, ultimately saving the animal’s life. Stem cells had grafted into the heart and formed new cardiac tissue. Additionally, the stem cell transplantation restarted cell cycle activity and halved the fibrosis that had been developing after the initial damage. Stem cell therapy also increased stamina and removed fluid buildup in the body, so characteristic in heart failure.
In future research, the scientists plan to characterize the cells more fully and to isolate the progenitors just by removing unwanted fat. These cells then can be moved to other locations in that same person for reconstructive purposes, to treat a variety of diseases, and also paradoxically to treat obesity and diabetes.
Identifying and isolating such a cell in humans would have profound therapeutic implications for disorders such as muscular dystrophy, injury and muscle wasting due to aging, disuse or disease.

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.