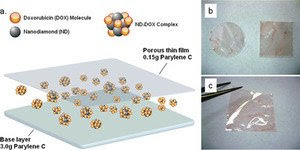
Nanodiamonds, a revolutionary class of nanomaterials, are 2 nanometers in diameter in single-particle form, and can be manipulated to form clusters with diameters in the 50-100nm range. This makes them ideal for drug delivery by shielding and slow releasing drugs trapped within the cluster of Diamond aggregates.
If a surgical oncologist, for example, was removing a tumor from the breast or brain, the device could be implanted in the affected area as part of the same surgery. This approach, which confines drug release to a specific location, could mitigate side effects and complications from other chemotherapy treatments.
Benefits
– Drugs can be slowly released over time (several months).
– nanodiamonds can trap nearly 5 times compared to conventional drug delivery
– the new system localizes the drugs to minimize and mitigate side effects
– nanodiamonds can be combined with a wide variety of drugs and RNA
To test the device’s drug release performance, the researchers used Doxorubicin, a chemotherapeutic used to treat many types of cancer. They found the drug slowly and consistently released from the embedded nanodiamond clusters for one month, with more Doxorubicin in reserve, indicating a more prolonged release (several months and longer) was possible. The device also avoided the “burst” or massive initial release of the drug, a common disadvantage with conventional therapy.
In addition to their large surface area, nanodiamonds have many other advantages that can be utilized in drug delivery. They can be functionalized with nearly any type of therapeutic. They can be suspended easily in water, which is important for biomedical applications. The nanodiamonds, each being four to six nanometers in diameter, are minimally invasive to cells, biocompatible and do not cause inflammation, a serious complication. And they are very scalable and can be produced in large quantities.
The architecture of the device is amenable to housing small molecule, protein, antibody or RNA- or DNA-based therapeutics. This gives the technology the potential to impact a range of treatment strategies where implanted, long-term drug release is needed.
“One of the most significant aspects of this work is that the fabrication procedures are highly scalable, meaning hundreds, or even thousands, of devices potentially could be manufactured in parallel and at low cost,” said Ho.
“The nanodiamonds are quite economical and have already been mass-produced as lubrication components for automobiles and for use in electronics,” added Robert Lam, a graduate student in Ho’s research group and the article’s lead author.
Because of the proven biocompatibility and massively parallel deposition capabilities of parylene, the researchers are engaged with pre-clinical trials of the nanodiamond-embedded parylene.
Taiwan has brought the cost of nanodiamonds down to about $300 for 10 milligrams.
FURTHER READING
The research paper, Nanodiamond-Embedded Microfilm Devices for Localized Chemotherapeutic Elution, is here
Dean Ho and his team are doing a lot of interesting work, which is described at his website.
More Nanoscale medicine by Dean Ho and team
Interrogation of cyto-regulatory networks via nanofabricated probe technology

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.