Researchers have created a new material that overcomes two of the major obstacles to solar power: it absorbs all the energy contained in visible sunlight, and generates electrons in a way that makes them 7 million times easier to capture.
Note: there is other work for capturing the infrared spectrum of sunlight.
Researchers and companies in the UK have delivered first generation single-junction cells with energy conversion efficiencies up to 12% for thermo-photovoltaic (TPV) cells. This compares to 9% from existing, commercially available devices. Increasing to 15% conversion of infrared energy to electricity is expected.
Ohio State University chemists and their colleagues combined electrically conductive plastic with metals including molybdenum and titanium to create the hybrid material.
Sunlight contains the entire spectrum of colors that can be seen with the naked eye — all the colors of the rainbow. What our eyes interpret as color are really different energy levels, or frequencies of light. Today’s solar cell materials can only capture a small range of frequencies, so they can only capture a small fraction of the energy contained in sunlight.
This new material is the first that can absorb all the energy contained in visible light at once.
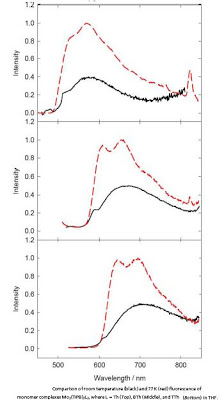
The molecules didn’t just fluoresce as some solar cell materials do. They phosphoresced as well. Both luminous effects are caused by a material absorbing and emitting energy, but phosphorescence lasts much longer.
To their surprise, the chemists found that the new material was emitting electrons in two different energy states — one called a singlet state, and the other a triplet state. Both energy states are useful for solar cell applications, and the triplet state lasts much longer than the singlet state.
Electrons in the singlet state stayed free for up to 12 picoseconds, or trillionths of a second — not unusual compared to some solar cell materials. But electrons in the triplet state stayed free 7 million times longer — up to 83 microseconds, or millionths of a second.
When they deposited the molecules in a thin film, similar to how they might be arranged in an actual solar cell, the triplet states lasted even longer: 200 microseconds.
“This long-lived excited state should allow us to better manipulate charge separation,” Chisholm said.
At this point, the material is years from commercial development, but he added that this experiment provides a proof of concept — that hybrid solar cell materials such as this one can offer unusual properties.
FURTHER READING
The actual paper:The remarkable influence of M2δ to thienyl π conjugation in oligothiophenes incorporating MM quadruple bonds
Abstract
Oligothiophenes incorporating MM quadruple bonds have been prepared from the reactions between Mo2(TiPB)4 (TiPB = 2,4,6-triisopropyl benzoate) and 3′,4′-dihexyl-2,2′-:5′,2″-terthiophene-5,5″-dicarboxylic acid. The oligomers of empirical formula Mo2(TiPB)2(O2C(Th)-C4(n-hexyl)2S-(Th)CO2) are soluble in THF and form thin films with spin-coating (Th = thiophene). The reactions between Mo2(TiPB)4 and 2-thienylcarboxylic acid (Th-H), 2,2′-bithiophene-5-carboxylic acid (BTh-H), and (2,2′:5′,2″-terthiophene)-5-carboxylic acid (TTh-H) yield compounds of formula trans-Mo2(TiPB)2L2, where L = Th, BTh, and TTh (the corresponding thienylcarboxylate), and these compounds are considered as models for the aforementioned oligomers. In all cases, the thienyl groups are substituted or coupled at the 2,5 positions. Based on the x-ray analysis, the molecular structure of trans-Mo2(TiPB)2(BTh)2 reveals an extended Lπ-M2δ-Lπ conjugation. Calculations of the electronic structures on model compounds, in which the TiPB are substituted by formate ligands, reveal that the HOMO is mainly attributed to the M2δ orbital, which is stabilized by back-bonding to one of the thienylcarboxylate π* combinations, and the LUMO is an in-phase combination of the thienylcarboxylate π* orbitals. The compounds and the oligomers are intensely colored due to M2δ–thienyl carboxylate π* charge transfer transitions that fall in the visible region of the spectrum. For the molybdenum complexes and their oligomers, the photophysical properties have been studied by steady-state absorption spectroscopy and emission spectroscopy, together with time-resolved emission and transient absorption for the determination of relaxation dynamics. Remarkably, THF solutions the molybdenum complexes show room-temperature dual emission, fluorescence and phosphorescence, originating mainly from 1MLCT and 3MM(δδ*) states, respectively. With increasing number of thienyl rings from 1 to 3, the observed lifetimes of the 1MLCT state increase from 4 to 12 ps, while the phosphorescence lifetimes are ≈80 μs. The oligomers show similar photophysical properties as the corresponding monomers in THF but have notably longer-lived triplet states, ≈200 μs in thin films. These results, when compared with metallated oligothiophenes of the later transition elements, reveal that M2δ–thienyl π conjugation leads to a very small energy gap between the 1MLCT and 3MLCT states of
Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.