New research from Harvard University, an unspecified factor in the blood of young mice can reverse signs of aging in the circulatory system of older ones. It’s not yet clear how these changes affect the animals’ overall health or longevity. But the research provides hope that some aspects of aging, such as the age-related decline in the ability to fight infection, might be avoidable.
Identifying those factors could lead to new strategies to boost resistance to infection, and perhaps a decrease in some cancers, she said. The findings, published today in the journal Nature, and which follow similar results with muscle stem cells, also suggest that the regenerative capacity of stem cells is highly influenced by their environment, which could have both positive and negative implications for regenerative medicine
One theory for aging is that our stem cells eventually wear out, thanks to intrinsic changes within the cells. While previous research supports this idea, findings from Wagers and others show that the age-related decline in stem cells is also influenced by external forces. For example, exposing skeletal muscle to blood-borne factors from young mice can restore the regenerative capacity of muscle stem cells.
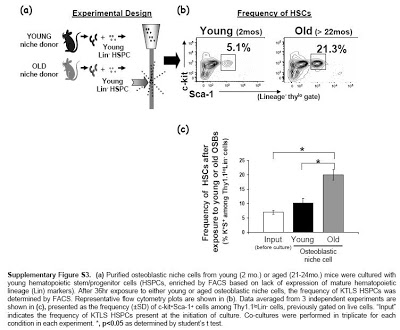
The regenerative power of young blood appears to be mediated by osteoblasts–bone-forming stem cells previously shown to play a role in regulating blood-forming stem cells. Researchers found that osteoblasts from old animals can make blood-forming stem cells from young mice act old. And conversely, surgically exposing old mice to young blood rejuvenates aged osteoblasts, restoring their capacity to properly regulate blood-forming stem cells.
Nature – Systemic signals regulate ageing and rejuvenation of blood stem cell niches
Ageing in multicellular organisms typically involves a progressive decline in cell replacement and repair processes, resulting in several physiological deficiencies, including inefficient muscle repair, reduced bone mass, and dysregulation of blood formation (haematopoiesis). Although defects in tissue-resident stem cells clearly contribute to these phenotypes, it is unclear to what extent they reflect stem cell intrinsic alterations or age-related changes in the stem cell supportive microenvironment, or niche. Here, using complementary in vivo and in vitro heterochronic models, we show that age-associated changes in stem cell supportive niche cells deregulate normal haematopoiesis by causing haematopoietic stem cell dysfunction. Furthermore, we find that age-dependent defects in niche cells are systemically regulated and can be reversed by exposure to a young circulation or by neutralization of the conserved longevity regulator, insulin-like growth factor-1, in the marrow microenvironment. Together, these results show a new and critical role for local and systemic factors in signalling age-related haematopoietic decline, and highlight a new model in which blood-borne factors in aged animals act through local niche cells to induce age-dependent disruption of stem cell function.
9 page pdf with supplemental information

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.