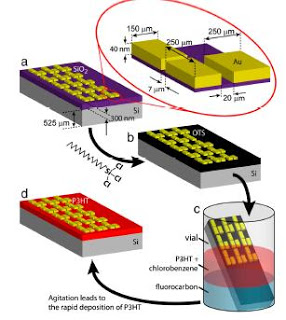
Flow process diagram. (A) An array of bottom-contact gold electrodes on SiO2∕Si and (Top Right) a close-up illustration of the electrode geometry.
(B) A self-adsorbed monolayer (SAM) of octadecyltrichlorosilane (OTS) is deposited on SiO2 by immersion into a 1% solution in toluene for 1 h. (C) The treated
electrode is immersed in a container with a solution of P3HT in chlorobenzene and a fluorocarbon such as Fluorinert FC-40®. This container is vigorously
agitated in order to form an emulsion of droplets that upon contact with the OTS SAM coalesce and lead to the deposition of a P3HT film. (D) The coated
electrode is taken out after 1 min of agitation and immediately annealed on top of a hot plate at 110 °C for 15 min.
A team of UCLA chemists and engineers has developed a new method for coating large surfaces with nanofiber thin films that are both transparent and electrically conductive. Their method involves the vigorous agitation of water, dense oil and polymer nanofibers. After this solution is sufficiently agitated it spreads over virtually any surface, creating a film.
“The beauty of this method lies in its simplicity and versatility,” said California NanoSystems Institute (CNSI) researcher Richard B. Kaner, a professor of chemistry and biochemistry and a professor of materials science and engineering at the UCLA Henry Samueli School of Engineering and Applied Science. “The materials used are inexpensive and recyclable, the process works on virtually any substrate, it produces a uniform thin film which grows in seconds and the entire thing can be done at room temperature.”
PNAS – Versatile solution for growing thin films of conducting polymers
The method employed for depositing nanostructures of conducting polymers dictates potential uses in a variety of applications such as organic solar cells, light-emitting diodes, electrochromics, and sensors. A simple and scalable film fabrication technique that allows reproducible control of thickness, and morphological homogeneity at the nanoscale, is an attractive option for industrial applications. Here we demonstrate that under the proper conditions of volume, doping, and polymer concentration, films consisting of monolayers of conducting polymer nanofibers such as polyaniline, polythiophene, and poly(3-hexylthiophene) can be produced in a matter of seconds. A thermodynamically driven solution-based process leads to the growth of transparent thin films of interfacially adsorbed nanofibers. High quality transparent thin films are deposited at ambient conditions on virtually any substrate. This inexpensive process uses solutions that are recyclable and affords a new technique in the field of conducting polymers for coating large substrate areas
Conducting polymers combine the flexibility and toughness of plastics with electrical properties. They have been proposed for applications ranging from printed electronic circuits to supercapacitors but have failed to gain widespread use because of difficulties processing them into films.
“Conducting polymers have enormous potential in electronics, and because this technique works with so many substrates, it can be used in a broad spectrum of applications, including organic solar cells, light-emitting diodes, smart glass and sensors,” said Yang Yang, a professor of materials science and engineering at the Samueli School of Engineering and Applied Science and faculty director of the Nano Renewable Energy Center at the CNSI.
One of the potential applications is smart, or switchable, glass that can change between states when an electric current is applied — for example, switching between see-through and opaque states to let light in or block it. The UCLA research group is applying the technique to other nanomaterials in addition to polymer nanofibers in the hopes of expanding the number of available applications.
The team’s solution-based technique, published in the peer-reviewed journal Proceedings of National Academy of Science, was discovered serendipitously when a transparent film of polymer spread up the walls of a container while nanofibers in water were being purified with chloroform.
When water and oil are mixed, a blend of droplets is formed, creating a water–oil interface that serves as an entry point for trapping polymer nanofibers at liquid–liquid interfaces. As droplets unite, a change in the concentration of blended solids at the water–oil interface leads to a difference in surface tension. Spreading up a glass wall occurs as result of an attempt to reduce the surface-tension difference. Directional fluid flow leads to a continuously conductive thin film comprised of a single monolayer of polymer nanofibers. The uniformity of the film surface is due to the particles being drawn out of the water–oil interface, sandwiched between two fluids of opposing surface tensions.
Development of the technology is occurring in collaboration with Fibron Technologies.
If you liked this article, please give it a quick review on ycombinator, or Reddit, or StumbleUpon. Thanks
Featured articles
Ocean Floor Gold and Copper
Ocean Floor Mining Company
var pubId=12340;
var siteId=12341;
var kadId=20815;
var kadwidth=300;
var kadheight=250;
var kadtype=1;

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.