Singularity Hub reports that stem cells can be used to give injured mice larger muscles for life.
Young mice with injured legs were given donor muscle stem cells from other young mice. Those injuries not only healed, but muscle mass increased 50% and muscle volume increased by an incredible 170%! Performance tests show the muscles were twice as strong as normal, and still above average when you control for size. Two years later, about the lifetime of a mouse, the legs were still bigger and stronger than normal, much to the scientists surprise.
* Instead of using stem cell transplants, they are working to use drugs that recreate the life long muscle mass increase.
* Olwin’s already tracking down the right signaling pathways, and has even had some success with dystrophic mice (that work is not yet published). Chemical signals seem better than stem cells for this application. By fooling muscles into taking the actions they would have taken had they received a transplant, Olwin is creating a possible systematic approach to treating muscle loss. You don’t need hundreds of micro-transplants, you just need one substance (or drug cocktail) that could be injected anywhere in the skeletal-muscular system.
* in studies with dystrophic mice (who are genetically induced to have MD) healthy well-connected muscle tissue did develop after stem cell transplant (that work has yet to be published).
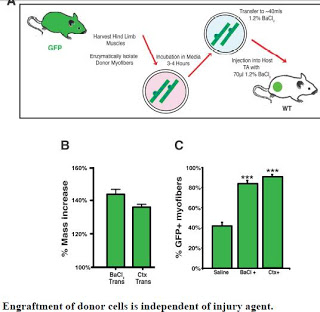
* injury stimulates the transplant
* The transplantation process itself is still being optimized. Olwin and colleagues found that you really had to get the donor cells when they were still attached to muscle fibers. Stripping them down and culturing them (as is done with many stem cell treatments) seems to decrease effectiveness. Remarkably, only about 10 to 50 stem cells were used for each mouse. That’s a surprisingly small number of cells. Olwin says that Hall and Chamberlain are already working on performing similar transplants in dogs. Clinical work in humans could be feasible in a few years.
Skeletal muscle is dynamic, adapting to environmental needs, continuously maintained, and capable of extensive regeneration. These hallmarks diminish with age, resulting in a loss of muscle mass, reduced regenerative capacity, and decreased functionality. Although the mechanisms responsible for this decline are unclear, complex changes within the local and systemic environment that lead to a reduction in regenerative capacity of skeletal muscle stem cells, termed satellite cells, are believed to be responsible. We demonstrate that engraftment of myofiber-associated satellite cells, coupled with an induced muscle injury, markedly alters the environment of young adult host muscle, eliciting a near-lifelong enhancement in muscle mass, stem cell number, and force generation. The abrogation of age-related atrophy appears to arise from an increased regenerative capacity of the donor stem cells, which expand to occupy both myonuclei in myofibers and the satellite cell niche. Further, these cells have extensive self-renewal capabilities, as demonstrated by serial transplantation. These near-lifelong, physiological changes suggest an approach for the amelioration of muscle atrophy and diminished function that arise with aging through myofiber-associated satellite cell transplantation.
11 pages of supplemental material
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks
Featured articles
Ocean Floor Gold and Copper
Ocean Floor Mining Company

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.