Researchers from UCLA and the Veterans Administration that was investigating how stress affects gastrointestinal function may have found a chemical compound that induces hair growth by blocking a stress-related hormone associated with hair loss — entirely by accident. The astressin-B peptide was responsible for the remarkable hair growth in the bald mice. Just one shot per day for five consecutive days maintained the effects for up to four months (16% of the 2 year lifespan of the mouse). Treatment of human hair loss could be achieved through the modulation of the stress hormone receptors, particularly hair loss related to chronic stress and aging. If it works in humans is appears to be a cure for baldness.
Researchers from UCLA and the Veterans Administration that was investigating how stress affects gastrointestinal function may have found a chemical compound that induces hair growth by blocking a stress-related hormone associated with hair loss — entirely by accident. The astressin-B peptide was responsible for the remarkable hair growth in the bald mice. Just one shot per day for five consecutive days maintained the effects for up to four months (16% of the 2 year lifespan of the mouse). Treatment of human hair loss could be achieved through the modulation of the stress hormone receptors, particularly hair loss related to chronic stress and aging. If it works in humans is appears to be a cure for baldness.
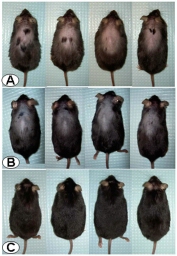
Corticotropin-releasing factor (CRF) signaling pathways are involved in the stress response, and there is growing evidence supporting hair growth inhibition of murine hair follicle in vivo upon stress exposure. We investigated whether the blockade of CRF receptors influences the development of hair loss in CRF over-expressing (OE)-mice that display phenotypes of Cushing’s syndrome and chronic stress, including alopecia. The non-selective CRF receptors antagonist, astressin-B (5 µg/mouse) injected peripherally once a day for 5 days in 4–9 months old CRF-OE alopecic mice induced pigmentation and hair re-growth that was largely retained for over 4 months. In young CRF-OE mice, astressin-B prevented the development of alopecia that occurred in saline-treated mice. Histological examination indicated that alopecic CRF-OE mice had hair follicle atrophy and that astressin-B revived the hair follicle from the telogen to anagen phase. However, astressin-B did not show any effect on the elevated plasma corticosterone levels and the increased weights of adrenal glands and visceral fat in CRF-OE mice. The selective CRF2 receptor antagonist, astressin2-B had moderate effect on pigmentation, but not on hair re-growth. The commercial drug for alopecia, minoxidil only showed partial effect on hair re-growth. These data support the existence of a key molecular switching mechanism triggered by blocking peripheral CRF receptors with an antagonist to reset hair growth in a mouse model of alopecia associated with chronic stress.
The researchers had been using mice that were genetically altered to overproduce a stress hormone called corticotrophin-releasing factor, or CRF. As these mice age, they lose hair and eventually become bald on their backs, making them visually distinct from their unaltered counterparts.
The Salk Institute researchers had developed the chemical compound, a peptide called astressin-B, and described its ability to block the action of CRF. Stenzel-Poore had created an animal model of chronic stress by altering the mice to overproduce CRF.
UCLA and VA researchers injected the astressin-B into the bald mice to observe how its CRF-blocking ability affected gastrointestinal tract function. The initial single injection had no effect, so the investigators continued the injections over five days to give the peptide a better chance of blocking the CRF receptors. They measured the inhibitory effects of this regimen on the stress-induced response in the colons of the mice and placed the animals back in their cages with their hairy counterparts.
About three months later, the investigators returned to these mice to conduct further gastrointestinal studies and found they couldn’t distinguish them from their unaltered brethren. They had regrown hair on their previously bald backs.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.