A team led by Dr Marc Pellegrini from the Walter and Eliza Hall Institute showed that a cell signaling hormone called interleukin-7 (IL-7) reinvigorates the immune response to chronic viral infection, allowing the host to completely clear virus. Their findings were released in today’s edition of the journal Cell. Dr Pellegrini said the research had provided excellent ideas for new therapies that could target and boost host immune cells to fight disease, rather than targeting the disease itself. “The findings could help to develop drugs that target some of these host molecules, such as SOCS-3, and turn them off for very short, defined periods of time to reinvigorate the T cells, allowing them to regroup to fight infection,” he said.
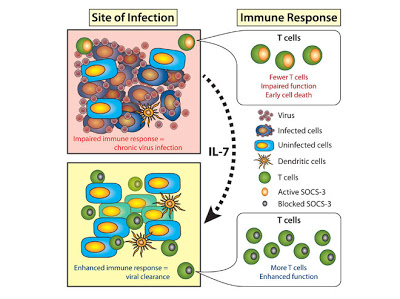
Understanding the factors that impede immune responses to persistent viruses is essential in designing therapies for HIV infection. Mice infected with LCMV clone-13 have persistent high-level viremia and a dysfunctional immune response. Interleukin-7, a cytokine that is critical for immune development and homeostasis, was used here to promote immunity toward clone-13, enabling elucidation of the inhibitory pathways underlying impaired antiviral immune response. Mechanistically, IL-7 downregulated a critical repressor of cytokine signaling, Socs3, resulting in amplified cytokine production, increased T cell effector function and numbers, and viral clearance. IL-7 enhanced thymic output to expand the naive T cell pool, including T cells that were not LCMV specific. Additionally, IL-7 promoted production of cytoprotective IL-22 that abrogated liver pathology. The IL-7-mediated effects were dependent on endogenous IL-6. These attributes of IL-7 have profound implications for its use as a therapeutic in the treatment of chronic viral diseases.
Dr Pellegrini, from the institute’s Infection and Immunity division, said the finding could lead to a cure for chronic viral infections such as HIV, hepatitis B and C, and bacterial infections such as tuberculosis, which are significant economic and global health burdens.
Current approaches to curing chronic infections tend to focus on generating a long-lived immune response to a specific disease. Dr Pellegrini, working with colleagues Mr Simon Preston and Mr Jesse Toe, and collaborators Professors Pamela Ohashi and Tak Mak from the Ontario Cancer Institute, argues that long-lived immune responses to chronic diseases are not always effective, and has instead concentrated on how the immune response can be manipulated to better fight infection.
“Viruses such as HIV and hepatitis B and C overwhelm the immune system, leading to establishment of chronic infections that are lifelong and incurable,” Dr Pellegrini said. “Despite tremendous efforts, long-lived immune responses for some of these viruses are ineffective, because the body is so overrun by virus that the immune system, in particular T cells, just give up trying to battle the infection. Some people have coined the phrase ‘immune exhaustion’ to explain the phenomenon. Our approach is to discover some of the mechanisms that cause this immune exhaustion, and manipulate host genes to see if we can boost the natural immune response in order to beat infection.”
The team investigated the role of IL-7, a naturally-occurring immune hormone, in a mouse model of HIV infection. IL-7 is a cytokine (cell signalling hormone) that plays a critical role in immune system development and maintenance.
“We found that IL-7 boosted the immune response in a pretty profound fashion, such that animals were able to gradually clear the virus without too much collateral tissue damage,” Dr Pellegrini said.
Further investigations revealed that, at the molecular level, IL-7 switched off a gene called SOCS-3.
“In an overwhelming infection, SOCS-3 becomes highly activated and suppresses the immune response, probably as a natural precaution to prevent ‘out-of-control’ responses that cause collateral damage to body tissue,” Dr Pellegrini said. “In the case of these overwhelming infections, the immune system effectively slams on the brakes too early, and the infection persists.”
Mr Preston, who worked on the SOCS-3 studies, said that switching off the SOCS-3 gene boosted the immune system and helped the animals to completely eliminate the infection.
“The key for us was figuring out that turning off SOCS-3 only really worked when it was within T cells,” Mr Preston said. “It allowed the immune response to boost the number of virus-specific T cells and have an immune response good enough to eliminate the virus without initiating an immune response that was too large and would make the animal sick.”
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.