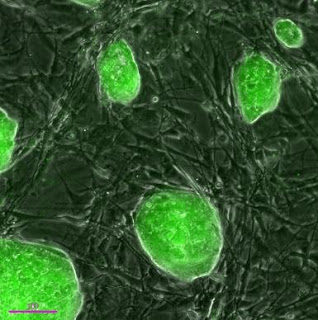
Mouse iPSCs generated using the microRNA method pioneered by the Morrisey Lab. The green fluorescence reveals expression of the Oct4 gene, which is a marker of pluripotent stem cells. Credit: Ed Morrisey, PhD, University of Pennsylvania School of Medicine
Researchers at the University of Pennsylvania School of Medicine have devised a totally new and far more efficient way of generating induced pluripotent stem cells (iPSCs), immature cells that are able to develop into several different types of cells or tissues in the body. The researchers used fibroblast cells, which are easily obtained from skin biopsies, and could be used to generate patient-specific iPSCs for drug screening and tissue regeneration.
iPSCs are typically generated from adult non-reproductive cells by expressing four different genes called transcription factors. The generation of iPSCs was first reported in 2006 by Shinya Yamanaka, and multiple groups have since reported the ability to generate these cells using some variations on the same four transcription factors.
The promise of this line of research is to one day efficiently generate patient-specific stem cells in order to study human disease as well as create a cellular “storehouse” to regenerate a person’s own cells, for example heart or liver cells. Despite this promise, generation of iPSCs is hampered by low efficiency, especially when using human cells.
“It’s a game changer,” says Edward Morrisey, PhD, professor in the Departments of Medicine and Cell and Developmental Biology and Scientific Director at the Penn Institute for Regenerative Medicine. “This is the first time we’ve been able to make induced pluripotent stem cells without the four transcription factors and increase the efficiency by 100-fold.” Morrisey led the study published this week in Cell Stem Cell.
“Generating induced pluripotent stem cells efficiently is paramount for their potential therapeutic use,” noted James Kiley, PhD, director of the National Heart, Lung, and Blood Institute’s Division of Lung Diseases. “This novel study is an important step forward in that direction and it will also advance research on stem cell biology in general.”
Before this procedure, which uses microRNAs instead of the four key transcription factor genes, for every 100,000 adult cells re-programmed, researchers were able to get a small handful of iPSCs, usually less than 20. Using the microRNA-mediated method, they have been able to generate approximately 10,000 induced pluripotent stem cells from every 100,000 adult human cells that they start with. MicroRNAs (miRNAs) are short RNA molecules that bind to complementary sequences on messenger RNAs to silence gene expression.
The Morrisey lab discovered this new approach through studies focusing on the role of microRNAs in lung development. This lab was working on a microRNA cluster called miR302/367, which plays an important role in lung endoderm progenitor development. This same microRNA cluster was reported to be expressed at high levels in embryonic stem cells, and iPSCs and microRNAs have been shown to alter cell phenotypes.
The investigators performed a simple experiment and expressed the microRNAs in mouse fibroblasts and were surprised to observe colonies that looked just like iPSCs. “We were very surprised that this worked the very first time we did the experiment,” says Morrisey. “We were also surprised that it worked much more efficiently than the transcription factor approach pioneered by Dr. Yamanaka.”
Since microRNAs act as repressors of protein expression, it seems likely that they repress the repressors of the four transcription factors and other factors important for maintaining the pluripotent-stem-cell state. However, exactly how the miRNAs work differently compared to the transcription factors in creating iPSCs will require further investigation.
The iPSCs generated by the microRNA method in the Morrisey lab are able to generate most, if not all, tissues in the developing mouse, including germ cells, eggs and sperm. The group is currently working with several collaborators to redifferentiate these iPSCs into cardiomyocytes, hematopoietic cells, and liver hepatocytes.
“We think this method will be very valuable in generating iPSCs from patient samples in a high-throughput manner” says Morrisey. microRNAs can also be introduced into cells using synthetically generated versions of miRNAs called mimics or precursors. These mimics can be easily introduced into cells at high levels, which should allow for a non-genetic method for efficiently generating iPSCs.
“The upshot is that we hope to be able to produce synthetic microRNAs to transform adult cells into induced pluripotent stem cells, which could eventually then be redifferentiated into other cell types, for example, liver, heart muscle or nerve cells” says Morrisey.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.