Stable two-dimensional networks of organic molecules are important components in various nanotechnology processes. However, producing these networks, which are only one atom thick, in high quality and with the greatest possible stability currently still poses a great challenge. Scientists from the Excellence Cluster Nanosystems Initiative Munich (NIM) have now successfully created just such networks made of boron acid molecules.
Researchers have developed a process by which they can build up high-quality polymer networks using boron acid components.
The “carpets” that the physicists are working on in their laboratory in the Deutsches Museum München consist of ordered two-dimensional structures created by self-organized boron acid molecules on a graphite surface. By eliminating water, the molecules bond together in a one-atom thick network held together solely by chemical bonds – a fact that makes this network very stable. The regular honey-comb-like arrangement of the molecules results in a nano-structured surface whose pores can be used, for instance, as stable forms for the production of metal nano-particles.
ACS Nano – Synthesis of Well-Ordered COF Monolayers: Surface Growth of Nanocrystalline Precursors versus Direct On-Surface Polycondensation
The molecular carpets also come in nearly perfect models; however, these are not very stable, unfortunately. In these models the bonds between the molecules are very weak – for instance hydrogen bridge bonds or van der Waals forces. The advantage of this variant is that faults in the regular structure are repaired during the self-organization process – bad bonds are dissolved so that proper bonds can form.
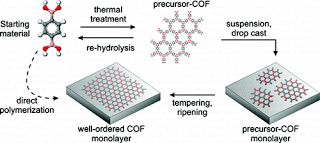
However, many applications call for molecular networks that are mechanically, thermally and/or chemically stable. Linking the molecules by means of strong chemical bonds can create such durable molecule carpets. The down side is that the unavoidable weaving mistakes can no longer be corrected due to the great bonding strength.
Markus Lackinger and his colleagues have now found a way to create a molecular carpet with stable covalent bonds without significant weaving mistakes. The method is based on a bonding reaction that creates a molecular carpet out of individual boron acid molecules. It is a condensation reaction in which water molecules are released. If bonding takes place at temperatures of a little over 100°C with only a small amount of water present, mistakes can be corrected during weaving. The result is the sought after magic carpet: molecules in a stable and well-ordered one-layer structure.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.