They are able to perceive light and even some shapes from the devices which were fitted behind the retina.
The men are part of a clinical trial carried out at the Oxford Eye Hospital and King’s College Hospital in London.
The two patients, Chris James and Robin Millar, lost their vision due to a condition known as retinitis pigmentosa, where the photoreceptor cells at the back of the eye gradually cease to function.
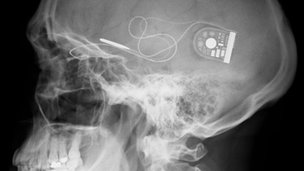
The wafer-thin, 3mm square microelectronic chip has 1,500 light-sensitive pixels which take over the function of the photoreceptor rods and cones.
The surgery involves placing it behind the retina from where a fine cable runs to a control unit under the skin behind the ear.
Retina Implant AG, the leading developer of subretinal implants for patients blinded by retinitis pigmentosa (RP), today announced that the first UK patients participating in the Company’s multi-centre trial have been successfully implanted. The UK trial is set to include 12 patients in total.
Retina Implant’s subretinal implant technology has been in clinical trials for more than six years. Patients involved in Retina Implant’s clinical trials have received a 3×3 mm2 microchip with 1,500 electrodes implanted below the retina. Results from the Company’s first human clinical trial published in Proceedings of the Royal Society B in November 2010 showed placement of the implant below the retina, in the macular region, provided optimum visual results allowing patients to recognise foreign objects and to read letters to form words. The second human clinical trial began in May 2010, in which patients were implanted with Retina Implant’s new wireless device in Germany, indicated even better visual acuity. The multi-centre phase of this trial was expanded in late 2011 and now includes two additional sites in Germany, and the UK, as well as a site in China. In fact, Retina Implant announced today the first of three patients to be implanted at the University of Hong Kong Eye Institute has regained useful sight after receiving Retina Implant’s microchip. Sites in Italy, Hungary and the U.S. are also under agreement to participate.
Data from the first nine patients implanted in Germany in this current trial indicate the best visual acuity to-date, with the majority of patients experiencing restoration of useful vision in daily life. The vast majority of patients are experiencing visual perception indoors and outdoors in both dim and bright environments. Additionally, patients have reported the ability to see objects 30 feet away and to read numbers on a pair of dice.
X-ray of skull showing position of chip with cable running to control unit
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.