Research group at Kiel University switches magnetism of individual molecules
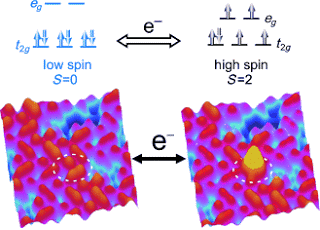
Using individual molecules instead of electronic or magnetic memory cells would revolutionise data storage technology, as molecular memories could be thousand-fold smaller. Scientists of Kiel University took a big step towards developing such molecular data storage. They succeeded in selectively switching on and off the magnetism of individual molecules, so-called spin-crossover complexes, by electrons. The interdisciplinary study is part of the Collaborative Research Centre 677 “Functions by Switching”, which is funded by the German Research Foundation (DFG). The results prove that it is technically possible to store information using molecules.
Single molecules of the iron(II) complex [Fe(bpz)2phen] (phen=1,10-phenanthroline, bpz=dihydrobis(pyrazolyl)borate) are reversibly switched between low spin and high spin by electron-induced spin crossover (ELIESST) in a bilayer on gold, as revealed by STM.
“In principle information may be stored in a single molecule. However, techniques that would make such an approach feasible are becoming available just now”, explains project leader Professor Richard Berndt of the Institute of Experimental and Applied Physics at Kiel University. Since the 1980s scientists are able to image individual molecules on surfaces with scanning tunnelling microscopes, he continues. Current research aims at controlling the characteristics of single molecules in order to facilitate future technical applications. The Collaborative Research Centre 677 “Functions by Switching” at Kiel University is a large-scale project engaged in such investigations, which aim at constructing molecular machines.
The current study is focused on the magnetism of molecules. Using a scanning tunnelling microscope Dr. Thiruvancheril Gopakumar, who carried out the study, was able to switch individual molecules between two magnetic states. Despite their dense packing in a molecular layer he was able to target individual molecules for switching. “Many research groups are striving to control the magnetic characteristics of molecules. Gopakumar’s studies have taken us one step ahead”, says Berndt.
The molecules (spin-crossover complexes) were synthesised at the Institute of Inorganic Chemistry at Kiel University. “Even though it took us a long time to find adequate molecules, we are very pleased with the outcome”, states Professor Felix Tuczek, head of the research group “Inorganic Molecular Chemistry”. The next step will be to adapt the molecules in a way that would allow scientists to switch them with light instead of electrons and at higher temperatures.
Structure of [Fe(bpz)2phen] in its LS state calculated with density functional theory at a tpssh/tzvp level (a)[1,2], along with wave functions of HOMO-1 (b), HOMO (c) and LUMO (d). A green triangle connects three pyrazole groups which, according to our model of the adsorption structure, point away from the surface. The thicker and shorter side of the triangle indicates two pyrazole groups of one dihydrobis(pyrazolyl)borate ligand. A high electron density is present at the pyrazole groups in the HOMO and HOMO-1 (marked by green dotted circles). The small energy difference between these orbitals and their broadening on a metal substrate explain the quasi-threefold symmetry of the molecule in STM images. e) STM image (V =−1.6 V, I = 50 pA) of a bilayer. The molecules in the second layer, which give rise to the STM image contrast, are expected to be in their LS state. Black and red triangles indicate molecules in adjacent rows. f) Corresponding top view of the proposed molecular arrangement in the second layer.
4 pages of supplemental information
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.