A graduate student and his team of researchers have turned the chemistry world on its ear by becoming the first ever to prove that tiny interlocked molecules can function inside solid materials, laying the important groundwork for the future creation of molecular machines.
“Until now, this has only ever been done in solution,” explained Chemistry & Biochemistry PhD student Nick Vukotic, lead author on a front page article recently published in the June issue of the journal Nature Chemistry. “We’re the first ones to put this into a solid state material.
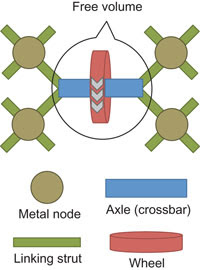
This schematic shows how the various elements assemble themselves into mechanically interlocked molecules.
Nature Chemistry – Metal–organic frameworks with dynamic interlocked components
The material Vukotic is referring to is UWDM-1, or University of Windsor Dynamic Material, a powdery substance that the team made which contains rotaxane molecules and binuclear copper centers. The rotaxane molecules, which resemble a wheel around the outside of an axle, were synthesised in their lab. The group found that heating of these rotaxane molecules with a copper source resulted in the formation of a crystalline material which contained structured arrangement of the rotaxane molecules, spaced out by the binuclear copper centers.
“Basically, they self-assemble in to this arrangement,” said Vukotic, who works under the tutelage of chemistry professor Steve Loeb. Other team members include professor Rob Schurko, and post-doctoral fellows Kristopher Harris and Kelong Zhu.
Heating the material causes the wheels to rapidly rotate around the axles, while cooling the material causes the wheels to stop, he said. The entire process can’t be viewed with a microscope, so the motion was confirmed in Dr. Schurko’s lab using a process called nuclear magnetic resonance spectroscopy.
“You can actually measure the motion and you can do it unambiguously by placing an isotopic tag on the ring,” explained Dr. Harris, who helped oversee that verification process.
Although the team admits their findings are still very much a proof of principle, they insist that molecules in solid materials can be manipulated to form switches and machines. This could be extremely significant and could find future applications in the fields of computer storage, data transfer or controlling the electronic properties of materials at the molecular level.
“Important metal organic frameworks like this are typically named after the university in which they were created,” Vukotic said of the material’s name. “It also emphasizes that this material is a prototype for other dynamic materials to come.”
Since the Nature Chemistry article appeared, a number of leading chemistry publications around the world have been reporting on the team’s findings, Dr. Loeb said.
“It’s a starting point,” said Loeb, who first imagined the concept about 10 years ago. “It’s a blueprint, but it’s the first demonstration of motion in a solid state. Normally when you make a new material you’re done. You can’t reorient molecules in a solid state.”
The dynamics of mechanically interlocked molecules such as rotaxanes and catenanes have been studied in solution as examples of rudimentary molecular switches and machines, but in this medium, the molecules eare randomly dispersed and their motion incoherent. As a strategy for achieving a higher level of molecular organization, we have constructed a metal–organic framework material using a [2]rotaxane as the organic linker and binuclear Cu(II) units as the nodes. Activation of the as-synthesized material creates a void space inside the rigid framework that allows the soft macrocyclic ring of the [2]rotaxane to rotate rapidly, unimpeded by neighbouring molecular components. Variable-temperature 13C and 2H solid-state NMR experiments are used to characterize the nature and rate of the dynamic processes occurring inside this unique material. These results provide a blueprint for the future creation of solid-state molecular switches and molecular machines based on mechanically interlocked molecules.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.