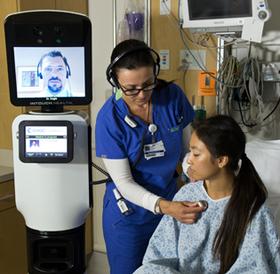
Shares of iRobot (Nasdaq:IRBT) soared to 10.27 percent to $23.28 as of 2 p.m. Thursday, Jan 24, 2013. The increase comes on the heels of the Bedford, Mass. company’s announcement that the remote presence robot developed by iRobot and its telemedicine partner, InTouch Health, has received 510(k) clearance by the by U.S. Food and Drug Administration for use in hospitals.
Six months ago iRobot launched the RP VITA (Remote Presence Virtual + Independent Telemedicine Assistant) at the InTouch Health 7th Annual Clinical Innovations Forum.
Daily Finance has a positive view of iRobot and the RP VITA
iRobot and InTouch Health are hard at work getting the remote-presence automatons into hospitals and health care facilities around the world. So far these initiatives seem to be paying off. InTouch has so far sold its telepresence robots to more than 400 hospitals. Meanwhile, iRobot’s Ava platform recently ranked among Information Week’s “10 Medical Robots That Could Change Healthcare.” The RP VITA robot can interface with diagnostic devices and electronic medical records (EMR) systems. The remote rig will eventually be able to navigate to specified target destinations autonomously.
The Vasteras Giraff is a mobile communication tool that enables the elderly to communicate with the outside world. It’s remote controlled, and it has wheels, a camera and a monitor. The Giraff provides two-way video calling similar to Skype. A caregiver can control the robot using a typical PC. To date, 42 Giraff robots have been delivered to seven European countries, and 20 more were recently built
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.