The flip of a single molecular switch helps create the mature neuronal connections that allow the brain to bridge the gap between adolescent impressionability and adult stability. Now Yale School of Medicine researchers have reversed the process, recreating a youthful brain that facilitated both learning and healing in the adult mouse.
Scientists have long known that the young and old brains are very different. Adolescent brains are more malleable or plastic, which allows them to learn languages more quickly than adults and speeds recovery from brain injuries. The comparative rigidity of the adult brain results in part from the function of a single gene that slows the rapid change in synaptic connections between neurons.
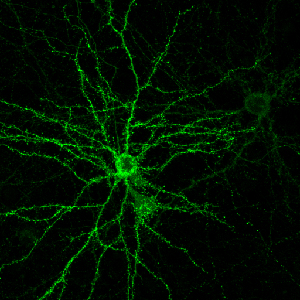
A cultured neuron with projecting dendrites studded with sites of communication between neurons, known as dendritic spines.
Journal Neuron – Anatomical Plasticity of Adult Brain Is Titrated by Nogo Receptor 1
By monitoring the synapses in living mice over weeks and months, Yale researchers have identified the key genetic switch for brain maturation a study released March 6 in the journal Neuron. The Nogo Receptor 1 gene is required to suppress high levels of plasticity in the adolescent brain and create the relatively quiescent levels of plasticity in adulthood. In mice without this gene, juvenile levels of brain plasticity persist throughout adulthood. When researchers blocked the function of this gene in old mice, they reset the old brain to adolescent levels of plasticity.
“These are the molecules the brain needs for the transition from adolescence to adulthood,” said Dr. Stephen Strittmatter. Vincent Coates Professor of Neurology, Professor of Neurobiology and senior author of the paper. “It suggests we can turn back the clock in the adult brain and recover from trauma the way kids recover.”
Rehabilitation after brain injuries like strokes requires that patients re-learn tasks such as moving a hand. Researchers found that adult mice lacking Nogo Receptor recovered from injury as quickly as adolescent mice and mastered new, complex motor tasks more quickly than adults with the receptor.
“This raises the potential that manipulating Nogo Receptor in humans might accelerate and magnify rehabilitation after brain injuries like strokes,” said Feras Akbik, Yale doctoral student who is first author of the study.
Researchers also showed that Nogo Receptor slows loss of memories. Mice without Nogo receptor lost stressful memories more quickly, suggesting that manipulating the receptor could help treat post-traumatic stress disorder.
“We know a lot about the early development of the brain,” Strittmatter said, “But we know amazingly little about what happens in the brain during late adolescence.”
Highlights
*Turnover of synaptic anatomy is accelerated in the brain of mice lacking NgR1
*Deletion of NgR1 in adult mice restores juvenile levels of anatomical plasticity
*NgR1 functions cell-autonomously to limit anatomical plasticity in vivo
*NgR1 expression limits the anatomical response to somatosensory input
Summary
Experience rearranges anatomical connectivity in the brain, but such plasticity is suppressed in adulthood. We examined the turnover of dendritic spines and axonal varicosities in the somatosensory cortex of mice lacking Nogo Receptor 1 (NgR1). Through adolescence, the anatomy and plasticity of ngr1 null mice are indistinguishable from control, but suppression of turnover after age 26 days fails to occur in ngr1−/− mice. Adolescent anatomical plasticity can be restored to 1-year-old mice by conditional deletion of ngr1. Suppression of anatomical dynamics by NgR1 is cell autonomous and is phenocopied by deletion of Nogo-A ligand. Whisker removal deprives the somatosensory cortex of experience-dependent input and reduces dendritic spine turnover in adult ngr1−/− mice to control levels, while an acutely enriched environment increases dendritic spine dynamics in control mice to the level of ngr1−/− mice in a standard environment. Thus, NgR1 determines the low set point for synaptic turnover in adult cerebral cortex.
SOURCE – Yale University, Journal Neuron
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.