Arxiv – Neural Dust: An Ultrasonic, Low Power Solution for Chronic Brain-Machine Interfaces
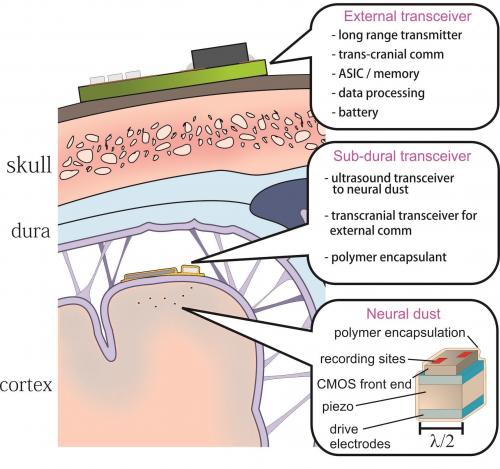
A major hurdle in brain-machine interfaces (BMI) is the lack of an implantable neural interface system that remains viable for a lifetime. This paper explores the fundamental system design trade-offs and ultimate size, power, and bandwidth scaling limits of neural recording systems built from low-power CMOS circuitry coupled with ultrasonic power delivery and backscatter communication. In particular, we propose an ultra-miniature as well as extremely compliant system that enables massive scaling in the number of neural recordings from the brain while providing a path towards truly chronic BMI.
These goals are achieved via two fundamental technology innovations:
1) thousands of 10 – 100 micron scale, free-floating, independent sensor nodes, or neural dust that detect and report local extracellular electrophysiological data, and
2) a sub-cranial interrogator that establishes power and communication links with the neural dust.
For 100 micron scale sensing nodes embedded 2 mm into the brain, ultrasonic power transmission can enable 7 % efficiency power links (-11.6 dB), resulting in a received power of 500 W with a 1 square mm interrogator, which is over 10 million times more than EM transmission at similar scale (40 pW). Extreme efficiency of ultrasonic transmission and CMOS front-ends can enable the scaling of the sensing nodes down to 10’s of microns
Dr. Michel Maharbiz: Neural dust system diagram showing the placement of ultrasonic interrogator under the skull and the independent neural dust sensing nodes dispersed throughout the brain.
Different materials or designs are needed to scale down further
Re-design of neural dust node
The scaling of both active and passive node implementations presented above is limited by the noise requirement of the front-end architectures, which is determined by the achievable differential signals between the electrodes. Decoupling the inherent tradeoff between the size of individual implants and the achievable SNR can improve the scaling of these implementations.
Re-thinking the design of neural dust can enhance its scalability. Since the trade-off derives directly not from the neural dust dimension, but from electrode separation, one approach may be to add very small footprint (1 – 5 micron wide) “tails” which position a single (or multiple) electrode relatively far (over 50 – 100 micron) from the base of the neural dust implant.
The analysis presented points to three major challenges in the realization of ultra-small, ultrasound-based neural recording systems. The first is the design and demonstration of front-ends suitable for operating within the extreme constraints of decreasing available power and decreasing SNR with scale. This could be addressed with a combination of CMOS process and design innovation as well as thinned, multi-substrate integration strategies.
The second challenge is the integration of extremely small piezoelectric transducers and CMOS electronics in a properly encapsulated package. The above discussion assumed the entire neural dust implant was encapsulated in an inert polymer or insulator film (a variety of such coatings are used routinely in neural recording devices; these include parylene, polyimide, silicon nitride and silicon dioxide, among others) while exposing two recording electrodes to the brain. The addition of “tails” as discussed above presents additional fabrication challenges.
The third challenge arises in the design and implementation of suitably sensitive sub-cranial transceivers which can operate at low power (to avoid heating between skull and brain). In addition to these three challenges, this paper does not discuss how to deliver neural dust nodes into the cortex. The most direct approach would be to implant them at the tips of fine-wire arrays similar to those already used for neural recording. Neural dust nodes would be fabricated or post-fab assembled on the tips of array shanks, held there by surface tension or resorbable layers; a recent result demonstrates a similar approach to implant untethered LEDs into neural tissue
Once inserted and free, the array shanks would be withdrawn, allowing the tissue to heal. Kinetic delivery might also be an option, but there is no existing data to evaluate what effect such a method would have on brain tissue or the devices themselves.
The trans-cranial transmitter design also introduces multi-interrogator, multi-node communication possibilities that will need to be developed in order to enable the large number of recording sites envisioned in this paper. Because the neural dust nodes are smaller than a wavelength, the reflected signals will be subject to diffraction. With multiple nodes embedded and sufficiently wide transceivers, this presents an interesting inverse problem of potential benefit in resolving signals from different nodes. An alternative approach to multi-node communication would be to fabricate nodes with a variety of resonant frequencies and use frequency discrimination (i.e., each dust transmits on its own frequency channel).
Lastly, neural dust nodes with aspect ratios close to 1:1:1 will not only couple energy into modes along the two axes perpendicular to the transmission axis, they will also re-radiate along those axes. This means nodes lying near each other on a “horizontal” plane (relative to the top surface of the cortex) may see inter-node signal mixing. This has interesting implications for node-to-node communication.
Lastly, one of the more compelling possibilities would be to harness the considerable volume of research that has gone into micro- and nanoelectromechanical RF resonators (which easily operate in the MHz range and thin-film piezoelectric transducers to produce devices with better power coupling as a function of scale, thus facilitating extremely small (10’s of microns) dust nodes. This remains an open opportunity.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.