USC Viterbi School of Engineering professor Chongwu Zhou and his research team have developed a silicon anode and a sulfur-based cathode with low fabrication cost and high electrode performance for rechargeable lithium-ion batteries
Researchers at the USC Viterbi School of Engineering have improved the performance and capacity of lithium batteries by developing better-performing, cheaper materials for use in anodes and cathodes (negative and positive electrodes, respectively).
Lithium-ion batteries are a popular type of rechargeable battery commonly found in portable electronics and electric or hybrid cars. Traditionally, lithium-ion batteries contain a graphite anode, but silicon has recently emerged as a promising anode substitute because it is the second most abundant element on earth and has a theoretical capacity of 3600 milliamp hours per gram (mAh/g), almost 10 times the capacity of graphite. The capacity of a lithium-ion battery is determined by how many lithium ions can be stored in the cathode and anode. Using silicon in the anode increases the battery’s capacity dramatically because one silicon atom can bond up to 3.75 lithium ions, whereas with a graphite anode six carbon atoms are needed for every lithium atom.
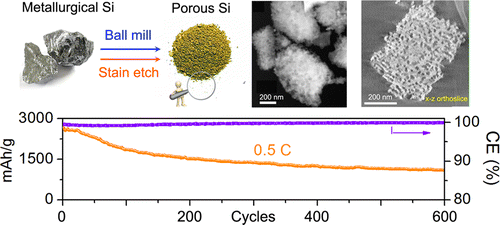
The USC Viterbi team developed a cost-effective (and therefore commercially viable) silicon anode with a stable capacity above 1100 mAh/g for extended 600 cycles, making their anode nearly three times more powerful and longer lasting than a typical commercial anode.
Up until recently, the successful implementation of silicon anodes in lithium-ion batteries faced one big hurdle: the severe pulverization of the electrode due to the volume expansion and retraction that occurs with the use of silicon. Last year, the same team led by USC Viterbi electrical engineering professor Chongwu Zhou developed a successful anode design using porous silicon nanowires that allowed the material to expand and contract without breaking, effectively solving the pulverization problem.
This solution yielded a new problem, however: the method of producing nanostructured silicon was prohibitively expensive for commercial adoption.
Undeterred, graduate student Mingyuan Ge and other members of Zhou’s team built on their previous work to develop a cost-efficient method of producing porous silicon particles through the simple and inexpensive methods of ball-milling and stain-etching.
“Our method of producing nanoporous silicon anodes is low-cost and scalable for mass production in industrial manufacturing, which makes silicon a promising anode material for the next generation of lithium-ion batteries,” said Zhou. “We believe it is the most promising approach to applying silicon anodes in lithium-ion batteries to improve capacity and performance.”
In addition, graduate student Jiepeng Rong and other team members developed a method of coating sulfur powder with graphene oxide to improve performance in lithium-sulfur batteries. Sulfur has been a promising cathode candidate for many years owing to its high theoretical capacity, which is over 10 times greater than that of traditional metal oxide or phosphate cathodes. Elemental sulfur is also abundant, cheap, and has low toxicity. However, the practical application of sulfur has been greatly hindered by challenges including poor conductivity and poor cyclability, meaning the battery loses power after each charge and dies after a lower number of recharges.
Their research proved that a graphene oxide coating over sulfur can solve both problems. Graphene oxide has unique properties such as high surface area, chemical stability, mechanical strength and flexibility, and is therefore commonly used to coat core materials in products like sensors or solar cells to improve their performance. The team’s graphene oxide coating improved the sulfur cathode’s capacity to 800 mAh/g for 1000 cycles of charge/discharge, which is over 5 times the capacity of commercial cathodes.
ABSTRACT- Large-Scale Fabrication, 3D Tomography, and Lithium-Ion Battery Application of Porous Silicon
Recently, silicon-based lithium-ion battery anodes have shown encouraging results, as they can offer high capacities and long cyclic lifetimes. The applications of this technology are largely impeded by the complicated and expensive approaches in producing Si with desired nanostructures. We report a cost-efficient method to produce nanoporous Si particles from metallurgical Si through ball-milling and inexpensive stain-etching. The porosity of porous Si is derived from particle’s three-dimensional reconstructions by scanning transmission electron microscopy (STEM) tomography, which shows the particles’ highly porous structure when etched under proper conditions. Nanoporous Si anodes with a reversible capacity of 2900 mAh/g was attained at a charging rate of 400 mA/g, and a stable capacity above 1100 mAh/g was retained for extended 600 cycles tested at 2000 mA/g. The synthetic route is low-cost and scalable for mass production, promising Si as a potential anode material for the next-generation lithium-ion batteries with enhanced capacity and energy density.
ABSTRACT –
A generic and facile method of coating graphene oxide (GO) on particles is reported, with sulfur/GO core–shell particles demonstrated as an example for lithium–sulfur (Li–S) battery application with superior performance. Particles of different diameters (ranging from 100 nm to 10 μm), geometries, and compositions (sulfur, silicon, and carbon) are successfully wrapped up by GO, by engineering the ionic strength in solutions. Importantly, our method does not involve any chemical reaction between GO and the wrapped particles, and therefore, it can be extended to vast kinds of functional particles. The applications of sulfur/GO core–shell particles as Li–S battery cathode materials are further investigated, and the results show that sulfur/GO exhibit significant improvements over bare sulfur particles without coating. Galvanic charge–discharge test using GO/sulfur particles shows a specific capacity of 800 mAh/g is retained after 1000 cycles at 1 A/g current rate if only the mass of sulfur is taken into calculation, and 400 mAh/g if the total mass of sulfur/GO is considered. Most importantly, the capacity decay over 1000 cycles is less than 0.02% per cycle. The coating method developed in this study is facile, robust, and versatile and is expected to have wide range of applications in improving the properties of particle materials.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.