Volkswagen spends roughly 13 billion Dollars on research and development each year.
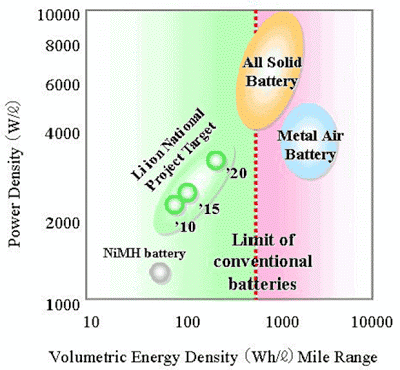
Winterkorn sees increasing the specific energy of lithium-ion cells to as much as 380 Wh/l will reduce driving range drawbacks. Lithium ion with a higher nickel content will get to even high energy density.
He sees great potential with solid-state batteries possibly boosting the range to as much as 700 kilometers (1,000 Wh/l).
Solid state lithium ion batteries replace the organic liquid electrolyte with a nonflammable and more reliable inorganic solid electrolyte (SE). This can provide higher energy density and simplify the battery design and improves safety and durability of the system.
Another matter is cost:
Lowering the price of battery cells to 100 euros ($120) per kilowatt hour would significantly increase the market potential of electric vehicles.
Toyota targets batteries better than gasoline which they call the Sakichi battery
Toyota established a research division to work on “revolutionary batteries.” At that time, the company talked about a “Sakichi battery,” named after Sakichi Toyoda, the inventor of Japan’s first power loom. He is sometimes referred to as the father of the Japanese industrial revolution. In 1925, Sakichi reportedly set oblamlut a (yet-to-be-claimed) prize of 1 million yen for the invention of a storage battery that would produce more energy than gasoline. Toyota’s goal is to make the Sakichi battery very durable and very quick to charge.
Toyota sees potential for the Magnesium Sulfur battery
Unlike lithium ions, which can carry only one electrical charge each, doubly charged magnesium ions shuttle two at a time — instantly multiplying the electrical energy that can be released for the same volume. Magnesium comes with its own challenge, however: whereas lithium zips through electrolytes and electrodes, magnesium with its two charges moves as if through treacle.
The US Department of Energy had 2011 estimates of different battery technology.
Lithium–sulphur (Li–S) technology, which uses extremely cheap materials and in theory can pack in five times more energy by weight than Li-ion (in practice, researchers suspect, it will probably be only twice as much).
One of Li–S’s main advantages, says chemical engineer Elton Cairns, is that it gets rid of the “dead weight” in a Li-ion battery. Inside a typical Li-ion cell, space is taken up by a layered graphite electrode that does little more than host lithium ions. These ions flow through a charge-carrying liquid electrolyte into a layered metal oxide electrode. As with all batteries, current is generated because electrons must flow around an outside circuit to balance the charges. To recharge the battery, a voltage is applied to reverse the electron flow, which also drives the lithium ions back.
In a Li–S battery, the graphite is replaced by a sliver of pure lithium metal that does double duty as both the electrode and the supplier of lithium ions: it shrinks as the battery runs, and reforms when the battery is recharged. And the metal oxide is replaced by cheaper, lighter sulphur that can really pack the lithium in: each sulphur atom bonds to two lithium atoms, whereas it takes more than one metal atom to bond to just one lithium. All of that creates a distinct weight and cost advantage for Li–S technology.
Lower cost batteries
Battery packs for electric cars cost more than $500 per kWh. “What’s holding back the mass acceptance of electric cars is really the price rather than the energy density,” he says. So IBM nanoscience Winfried Wilcke now favours a cheaper breathing battery based on sodium. Theory predicts that sodium–oxygen (Na–O) batteries could provide only half the energy density of Li–O, but that is still five times better than Li-ion batteries. And sodium is cheaper than lithium, so Na–O might, Wilcke hopes, get closer to the $100 per kWh goal that the JCESR and others have set for affordability.
SOURCES: Volkswagen, Toyota, Nature Journal

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.