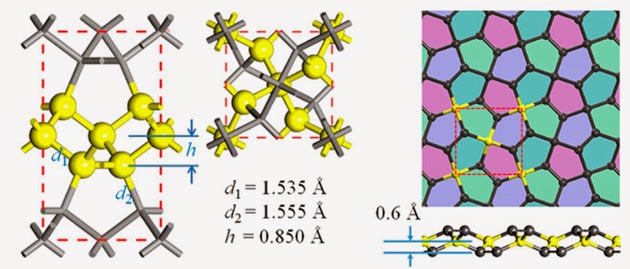
Jon Evans at Chemistry World reports that scientists in China have proposed the existence of a novel 2D allotrope of carbon made up of pentagons, which they have dubbed ‘penta-graphene’. Like graphene, penta-graphene consists of an atom-thick sheet of carbon atoms, but the atoms are arranged in a repeated pentagon pattern, potentially giving it some intriguing and useful physical properties.
In conventional graphene, the carbon atoms make a repeated hexagon pattern like chicken wire. Spherical C60 buckyballs are made up of both hexagons and pentagons, but scientists have so far only been able to synthesise a single carbon nanomaterial made exclusively of pentagons, in the form of a C20 cage. Nevertheless, this does raise the possibility of making other pentagon-based allotropes.
Proceedings of the National Academy of Science – Penta-graphene: A new carbon allotrope
Useful properties
The team found that not only should a pentagon-containing version of graphene be fairly stable, it should also be stronger than conventional graphene and be able to withstand higher temperatures, up to 730°C. It would also be a natural semiconductor, unlike conventional graphene, which is a highly efficient conductor and has to be chemically modified to turn it into a semiconductor. This could potentially prove useful for manufacturing carbon nanotubes (CNTs), as current CNT synthesis processes produce a mixture of metallic and semiconducting CNTs, depending on the orientation of the hexagons when a graphene sheet is rolled up. In contrast, rolling up a sheet of penta-graphene should only produce semiconducting CNTs.
These properties suggest a whole host of potential applications for penta-graphene, from computer circuits to body-armour. ‘[It] may have important applications in nano-electronics, biomaterials, tension-activatable substrates and nano-auxetic materials, and may even find applications in space technologies,’ Wang suggests.
Realising these applications will obviously require penta-graphene to be synthesised, which Wang thinks can be done by peeling single sheets from a layered material known as T12-carbon, although this arrangement of carbon is also purely theoretical at the moment. ‘We are looking for some experimental groups in China that are good at synthesising new materials,’ says Wang. ‘Penta-graphene is energetically more stable than C20 [and so] we hope that it will not be so difficult to synthesise.
Significance
Carbon has many faces––from diamond and graphite to graphene, nanotube, and fullerenes. Whereas hexagons are the primary building blocks of many of these materials, except for C20 fullerene, carbon structures made exclusively of pentagons are not known. Because many of the exotic properties of carbon are associated with their unique structures, some fundamental questions arise: Is it possible to have materials made exclusively of carbon pentagons and if so will they be stable and have unusual properties? Based on extensive analyses and simulations we show that penta-graphene, composed of only carbon pentagons and resembling Cairo pentagonal tiling, is dynamically, thermally, and mechanically stable. It exhibits negative Poisson’s ratio, a large band gap, and an ultrahigh mechanical strength.
Abstract
A 2D metastable carbon allotrope, penta-graphene, composed entirely of carbon pentagons and resembling the Cairo pentagonal tiling, is proposed. State-of-the-art theoretical calculations confirm that the new carbon polymorph is not only dynamically and mechanically stable, but also can withstand temperatures as high as 1000 K. Due to its unique atomic configuration, penta-graphene has an unusual negative Poisson’s ratio and ultrahigh ideal strength that can even outperform graphene. Furthermore, unlike graphene that needs to be functionalized for opening a band gap, penta-graphene possesses an intrinsic quasi-direct band gap as large as 3.25 eV, close to that of ZnO and GaN. Equally important, penta-graphene can be exfoliated from T12-carbon. When rolled up, it can form pentagon-based nanotubes which are semiconducting, regardless of their chirality. When stacked in different patterns, stable 3D twin structures of T12-carbon are generated with band gaps even larger than that of T12-carbon. The versatility of penta-graphene and its derivatives are expected to have broad applications in nanoelectronics and nanomechanics.
SOURCES- Chemistry World, PNAS

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.