A gene drive is a piece of “selfish” DNA that can spread rapidly through a population. But fears that engineered gene drives could spread out of control may be exaggerated as there are flaws in the existing designs that mean they will not last long in the wild. Modified versions of gene drive are in the works that might just deliver on the technology’s enormous potential to do good or bad.
Most plants and animals have matching pairs of chromosomes, but pass down only one of each pair to an offspring – the other comes from the other parent. This means that if you add a piece of DNA to one chromosome, normally only half the offspring will inherit it.
Gene drives cheat by “copying and pasting” themselves to the other chromosome too, meaning all offspring inherit them and they can spread rapidly throughout a population. This happens even when the new DNA confers a disadvantage to the offspring – which is the very reason the technology is so beneficial to us.
Engineer a gene drive to spread genes that stop mosquitoes carrying malaria, for instance, and you could eradicate the disease once and for all. A similar technique could also win the war on Zika virus.
However, one fear over this research is that experimental gene drives could escape from labs and begin to spread – perhaps with disastrous consequences, such as driving a beneficial species to extinction. Another fear is that if gene drives are deliberately released and turn out to have unintended consequences, there may be little we can do to stop them spreading.
In practice though, resistance to a gene drive often evolves in just a few generations, meaning these fears may be unfounded – for now anyway.
BioRxIV – Evolutionary dynamics of CRISPR gene drives
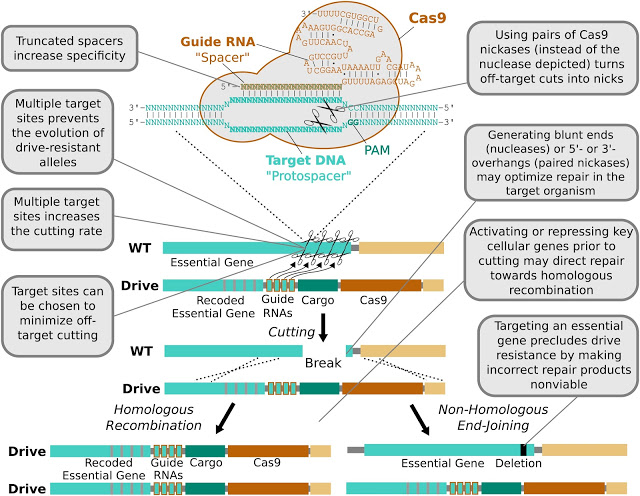
For CRISPR-based gene drives to copy and paste themselves from one chromosome to another, they have to recognize a specific target DNA sequence, cut it, and splice a copy of themselves into the gap. But natural variation means that some organisms may have chromosomes in which the target DNA is different or missing. These naturally resistant chromosomes can spread through a population, making them immune to a specific gene drive.
Second generation gene drive are targeting DNA sites that vary little between individuals.
The copy-and-paste process can also fail. Instead of a copy of a gene drive being pasted in after the target DNA is cut, the severed ends are sometimes crudely stuck back introducing mutations in the target DNA. In other words, even when naturally resistant chromosomes don’t exist, CRISPR gene drives can generate them.
According to a model developed by George Church’s team at Harvard University, this means that while existing CRISPR gene drives can spread rapidly at first, resistance will appear and the gene drive will disappear after a hundred generations or so.
But they have a cunning plan: to make the target DNA part of a crucial gene and to cut this gene in several places, not just one. By doing this, the crucial gene is destroyed. The gene drive includes the sequences needed to repair the gene. This means that if the copy and paste process works perfectly, the crucial gene is repaired along with the gene drive.
If the process goes awry and the ends are just stuck together, however, the gene containing the target DNA will be wrecked. Because it is crucial to the organism’s survival, the chromosome carrying it will not spread despite becoming resistant to the gene drive.
Abstract
The alteration of wild populations has been discussed as a solution to a number of humanity’s most pressing ecological and public health concerns. Enabled by the recent revolution in genome editing, CRISPR gene drives, selfish genetic elements which can spread through populations even if they confer no advantage to their host organism, are rapidly emerging as the most promising approach. But before real-world applications are considered, it is imperative to develop a clear understanding of the outcomes of drive release in nature. Toward this aim, we mathematically study the evolutionary dynamics of CRISPR gene drives. We demonstrate that the emergence of drive-resistant alleles presents a major challenge to previously reported constructs, and we show that an alternative design which selects against resistant alleles greatly improves evolutionary stability. We discuss all results in the context of CRISPR technology and provide insights which inform the engineering of practical gene drive systems.
SOURCES- New Scientist, BioRXIV

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.