A research team at the University of Washington has harnessed complex computational methods to design customized proteins that can self-assemble into 120-subunit “icosahedral” structures inside living cells—the biggest, self-booting, intracellular protein nanocages ever made. The breakthrough offers a potential solution to a pressing scientific challenge: how to safely and efficiently deliver to cells new and emerging biomedical treatments such as DNA vaccines and therapeutic interfering particles.
The work, funded by DARPA in a lead-up to the new INTERfering and Co-Evolving Prevention and Therapy (INTERCEPT) program, “opens the door to a new generation of genetically programmable protein-based molecular machines,” the researchers report in this week’s issue of the journal Science.
The unified atomic mass unit or dalton is the standard unit that is used for indicating mass on an atomic or molecular scale (atomic mass). One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol. It is defined as one twelfth of the mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state.
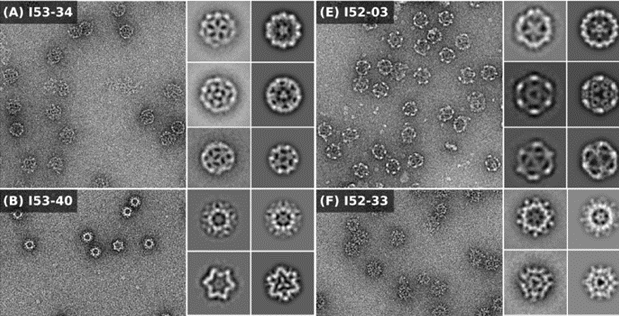
Characterization of the designed protein cages using electron microscopy. All of the raw micrographs are shown to scale relative to the 30-nanometer scale bar in panel H. (Image: University of Washington via Science)
Science – Accurate design of megadalton-scale two-component icosahedral protein complexes
Anyone familiar with the role-playing games Dungeons and Dragons and Munchkin need only picture the 20-sided die to understand what an organic, icosahedral cargo container looks like—symmetrical, triangle-shaped panels folded evenly on each side. Unlike a die that can be held in your hand, however, these creations are the size of small viruses and are designed to interact with cells in the same way viruses might—that is, by delivering their caged contents into a cell, albeit in this case with positive, customizable outcomes. Also, whereas dice are produced in molds on a factory assembly line, these nanocages build themselves inside cells, following with atomic precision instructions written in genetic code.
Nature provides many examples of self- and co-assembling protein-based molecular structures like icosahedral protein cages. They serve as scaffolds, enzymes, and compartments for biochemical reactions in the body, and they form virus capsids—the geodesic-like protein enclosures that protect viral genomes from the human body’s immune system and facilitate their entry into cells.
“Viruses offer researchers many lessons on ways to access the body and use the body’s resources for their own purposes. DARPA is studying how to apply those tricks to the challenge of overcoming infectious disease,” said Jim Gimlett, the DARPA program manager overseeing the University of Washington effort and the INTERCEPT program. Among other goals, that program seeks to deliver into cells protein snippets that can co-evolve with viruses and provide ongoing immune protection even as viruses mutate and change.
Viruses consist of two main parts: genetic material (DNA or RNA) surrounded by a protein shell. To reproduce, a virus first attaches itself to a cell within a host organism. This occurs when a virion protein on the virus binds to a complementary molecule on the surface of the cell. The virus then enters the cell and releases its genetic material inside. These viral genes hijack the cell’s biological machinery, forcing the cell to generate new copies of the viral genome and shell proteins. While still inside the host cell, those freshly minted viral genomes and protein shells assemble into new viruses, which eventually burst from the cell and disperse to infect others.
DARPA’s focus is on the protein shells. If scientists could design customized shells and program them to embrace specific payloads and replicate within the body, they could open new pathways for personalized medicines and therapies. However, the universe of proteins is large, and the combinations of ways in which those proteins can—based on electromagnetic charges and other factors—link to one another and spontaneously fold within three-dimensional space is devilishly complex.
When designing a protein assembly process, the nature of the selected proteins and how they fold will determine if that creation can bind to particular cell receptors and whether it can accommodate a desired cargo such as small molecules, antibodies, nucleic acids, polymers, or other proteins. “Protein folding can be compared to a 3-D puzzle with thousands of pieces and an astronomical number of possible solutions, but only one of those solutions might be right for a particular need,” Gimlett said.
The University of Washington’s latest research used combinatorial computations to consider hundreds of thousands of possible protein combinations and then characterize in detail a few hundred of those designs. Then the team inserted genes encoding the desired protein sequences into E. coli bacteria to test how the instructions would be carried out within living cells. Ten of the designs resulted in assemblies within those bacteria similar to the intended three-dimensional configurations. In fact, tests conducted using small-angle X-ray scattering and negative-stain electron microscopy revealed that these cage-like structures had self-assembled with near-perfect, atomic-scale precision.
Icosahedra possess the highest possible symmetry of any polyhedron in three-dimensional space, and thus generate the maximum enclosed volume for packaging cargo among symmetric assemblies of polymers. The accurate self-assembly of a 120-subunit icosahedral protein cage is a five-fold improvement over what had been, until recently, the previous record for such a structure: a 24-subunit, two-component tetrahedron. Just last month, the Seattle team announced that it had broken that record by designing a hyperstable, self-assembling 60-subunit icosahedron, research that was featured in the journal Nature. In their latest paper, the researchers note that the 120-subunit assembly either hasn’t evolved in nature or has yet to be discovered.
“This construct’s generous capacity, and the accuracy with which it builds itself, bode well for the field of biomolecular engineering,” Gimlett said. “I look forward to seeing some of the innovative applications that will surely emerge with the arrival of this robust new platform.”
Designed to assemble
Symmetric macromolecular structures that form cages, such as viral capsids, have inspired protein engineering. Bale et al. used pairwise combinations of dimeric, trimeric, or pentameric building blocks to design two-component, 120-subunit protein complexes with three distinct icosahedral architectures. The capsid-like nanostructures are large enough to hold nucleic acids or other proteins, and because they have two components, the assembly of cargoes such as drugs and vaccines can be done in a controlled way.
Abstract
Nature provides many examples of self- and co-assembling protein-based molecular machines, including icosahedral protein cages that serve as scaffolds, enzymes, and compartments for essential biochemical reactions and icosahedral virus capsids, which encapsidate and protect viral genomes and mediate entry into host cells. Inspired by these natural materials, we report the computational design and experimental characterization of co-assembling, two-component, 120-subunit icosahedral protein nanostructures with molecular weights (1.8 to 2.8 megadaltons) and dimensions (24 to 40 nanometers in diameter) comparable to those of small viral capsids. Electron microscopy, small-angle x-ray scattering, and x-ray crystallography show that 10 designs spanning three distinct icosahedral architectures form materials closely matching the design models. In vitro assembly of icosahedral complexes from independently purified components occurs rapidly, at rates comparable to those of viral capsids, and enables controlled packaging of molecular cargo through charge complementarity. The ability to design megadalton-scale materials with atomic-level accuracy and controllable assembly opens the door to a new generation of genetically programmable protein-based molecular machines.
SOURCES – DARPA, Science, wikipedia

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.