A team of researchers at Cornell University has built and run a simulation that showed prebiotic reactions could possibly occur on the surface of one of Saturn’s moons, Titan, suggesting the possibility of life evolving in a place where it is too cold for water to be a factor. The team describes the simulation they created in response to the discovery (by the Huygens probe) that polymers such as polyimine might have already developed on the moon’s surface.
Recent evidence has suggested that some chemical reactions could lead to types of life forms where there is no water, which further suggests that it may exist outside of what is now considered the habitual zone.
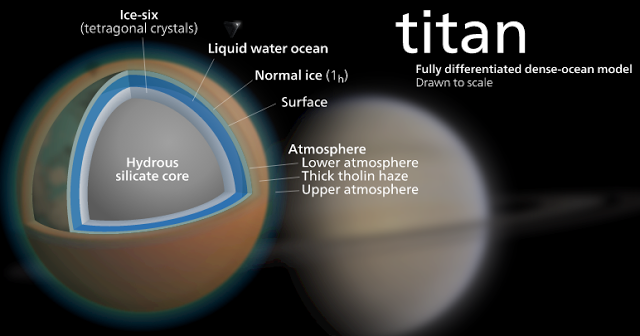
For life to come about in such places, researchers reason, there would likely need to be some sort of action going on—and that is why there has been so much focus on Titan; it is the only object in our solar system, besides Earth, that has both rainfall and erosion due to liquid movemen
Significance
Titan is the only place in the solar system, except Earth, where rainfall and seasonally flowing liquids erode the landscape. Whereas the surface pressure is similar to that of Earth, the temperature is extremely low and the dominant liquids are methane and ethane. This makes Titan a test case for exploring the environmental limits of prebiotic chemistry and addressing the question of whether life can develop without water. Experimental and observational data suggest that hydrogen cyanide, the most abundant hydrogen-bonding molecule in Titan’s atmosphere, may polymerize on the surface to polyimine. Using quantum mechanical calculations, we show that polyimine has interesting electronic and structural properties that could potentially facilitate prebiotic chemistry under cryogenic conditions akin to those on Titan.
Abstract
The chemistry of hydrogen cyanide (HCN) is believed to be central to the origin of life question. Contradictions between Cassini–Huygens mission measurements of the atmosphere and the surface of Saturn’s moon Titan suggest that HCN-based polymers may have formed on the surface from products of atmospheric chemistry. This makes Titan a valuable “natural laboratory” for exploring potential nonterrestrial forms of prebiotic chemistry. We have used theoretical calculations to investigate the chain conformations of polyimine (pI), a polymer identified as one major component of polymerized HCN in laboratory experiments. Thanks to its flexible backbone, the polymer can exist in several different polymorphs, which are relatively close in energy. The electronic and structural variability among them is extraordinary. The band gap changes over a 3-eV range when moving from a planar sheet-like structure to increasingly coiled conformations. The primary photon absorption is predicted to occur in a window of relative transparency in Titan’s atmosphere, indicating that pI could be photochemically active and drive chemistry on the surface. The thermodynamics for adding and removing HCN from pI under Titan conditions suggests that such dynamics is plausible, provided that catalysis or photochemistry is available to sufficiently lower reaction barriers. We speculate that the directionality of pI’s intermolecular and intramolecular =N–H…N hydrogen bonds may drive the formation of partially ordered structures, some of which may synergize with photon absorption and act catalytically. Future detailed studies on proposed mechanisms and the solubility and density of the polymers will aid in the design of future missions to Titan
SOURCES- PNAS , Phys.org, Cornell University

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.


Comments are closed.