Fightaging notes the importance of the next five years for antiaging therapy.
Donate to SENS research antiaging here
The next five years are critical precisely because the first few startup companies to work on the first rejuvenation therapies following the SENS model of damage repair will succeed or fail in this short span of time. The most important of these companies are probably Oisin Biotechnologies and UNITY Biotechnology, both working on senescent cell clearance. They have what looks like the best chance of success given the present state of the science, and are already well underway. Technical success does not necessarily translate to rapid clinical availability in medicine, however. You only have to look at Pentraxin Therapeutics and their work on transthyretin amyloid clearance to see that: they have been locked into a development program with GlaxoSmithKline that took six years to get to a small clinical trial, and there is no sign that this will move any faster following the success of that trial, or that it will be made available for anyone other than late stage amyloidosis patients. Clearance of transthyretin amyloid should be undergone by every human being every few years after the age of 40, given that buildup of this form of amyloid contributes to heart disease and a range of other conditions – but that development group simply isn’t heading in that direction. It is one thing to catch the interest of Big Pharma, another thing entirely to make them work rapidly, or to agree with the vision of treating aging as a medical condition.
Thus there is a very large difference between (a) a world in which companies conservatively sell to Big Pharma or raise funds themselves to creep through the regulatory process to gain approval for use with a tiny number of patients in the late stages of aging, and (b) a world in which the first destination is clinical availability via medical tourism in regulatory regions where only safety must be demonstrated, and anyone can walk in and undergo treatment. Stem cell medicine would be nowhere near as far along without the decade of its widespread availability outside the US and Europe. I am very much in favor of a similar progression of availability and development for the range of potentially useful human gene therapies that will be developed in the years ahead, and for the first SENS rejuvenation technologies, such as senescent cell clearance.
Nonetheless, whether or not the outcome is much delayed availability of therapies, success in building a company based on SENS therapies is a very important step. It will in some cases, as for Oisin Biotechnologies, bring significant funding for other lines of SENS research as various advocates and the SENS Research Foundation are early investors. More importantly, success in clinical development of a treatment that meaningfully addresses easily measured metrics of aging after one set of treatments – metrics such as the epigenetic clock based on DNA methylation, or inflammation, or skin elasticity, or blood vessel elasticity, and so on – will be widely noted. That will go a long way towards opening many doors to much larger sources of funding. Either this happens soon, for the companies already under way, or they will fail, possibly damaging the view of SENS even should that failure happen for reasons unrelated to the technical aspects of the work. Failure will push back ultimate success in the medical control of aging for years, and that has an enormous cost associated with it: tens of millions of lives lost, and hundreds of millions suffering due to age-related conditions that might otherwise have been turned back. The SENS Research Foundation staff realize this well, and hence their focus on Project|21, launched earlier this year.
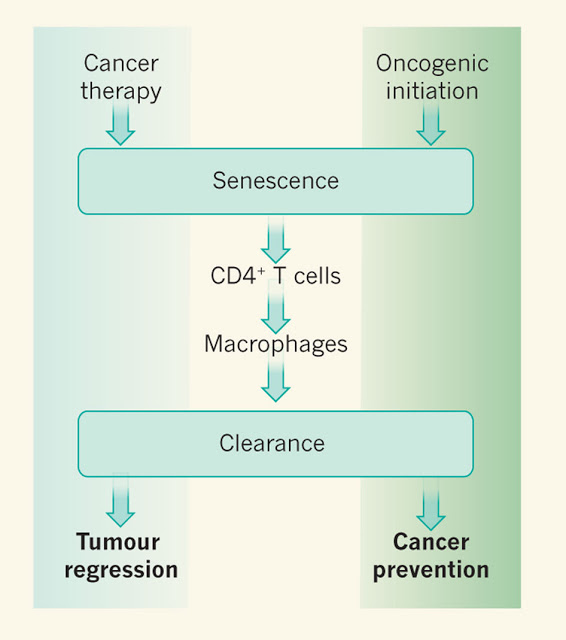
(2011) Nature – Cancer: Final act of senescence
Oisín is developing a genetically-targeted intervention to clear senescent cells. As a recent study has shown, clearing senescent cells both reduces negative effects of aging pathologies and also extends median lifespan and survival.
Donate to SENS research antiaging here
SOURCES – Oisin Biotechnology, SENS, Fight Aging, Nature

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.