1. being more-effective tumor responses.
2. the targeted nature of CRISPR-mediated CAR integration into the genome might “prove safer than random integration, which carries the potential risk of generating a harmful mutation,” Dr. Maus wrote.
3. It could enable off-the-shelf CAR T cells to be made that need not come from a patient’s own T cells. This would enable easier and cheaper manufacture of CAR T cells.
New gene-editing technologies will likely lead to rapid improvement in antigen-targeted T-cell immunotherapies for cancer.
David Edgell, an associate professor of biochemistry at the University of Western Ontario, thinks CRISPR treatments could be available within the next two to three years, with modified T-cells used to treat some types of cancer (there are already clinical trials for lung cancer in China, and a similar one slated to take place at the University of Pennsylvania was approved last June by the National Institutes of Health
CAR T cells created with CRISPR were less likely to stop recognizing and attacking tumor cells after a certain time point, a phenomenon researchers call “exhaustion.”
Based on three measures of exhaustion, less than 2% of CRISPR-created T cells showed signs of exhaustion, compared with up to half of conventionally engineered CAR T cells.
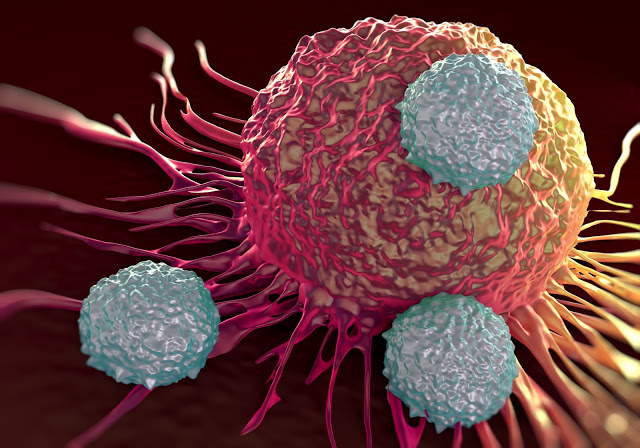
The cells were modified to express proteins on their surfaces called chimeric antigen receptors (CARs), which enabled the cells to recognize and attack cancer cells that expressed the corresponding antigen.
In experiments with the mice, immune cells that had been engineered to express CARs using CRISPR were more effective at killing tumor cells than immune cells engineered using conventional methods.
Nature – Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.