CRISPR DNA editing is allowing correction of many diseases. But many other diseases are linked to RNA. Biotech startup Locana is using the power of CRISPR to develop treatments for disease caused by RNA.
They use a technique for tracking RNA in live cells called RNA-targeting Cas9 (RCas9). In a new study, published August 10 in Cell, the team takes RCas9 a step further: they use the technique to correct molecular mistakes that lead to microsatellite repeat expansion diseases, which include myotonic dystrophy types 1 and 2, the most common form of hereditary ALS, and Huntington’s disease.
“This is exciting because we’re not only targeting the root cause of diseases for which there are no current therapies to delay progression, but we’ve re-engineered the CRISPR-Cas9 system in a way that’s feasible to deliver it to specific tissues via a viral vector,” said senior author Gene Yeo, PhD, professor of cellular and molecular medicine at UC San Diego School of Medicine.
While DNA is like the architect’s blueprint for a cell, RNA is the engineer’s interpretation of the blueprint. In the central dogma of life, genes encoded in DNA in the nucleus are transcribed into RNA and RNAs carry the message out into the cytoplasm, where they are translated to make proteins.
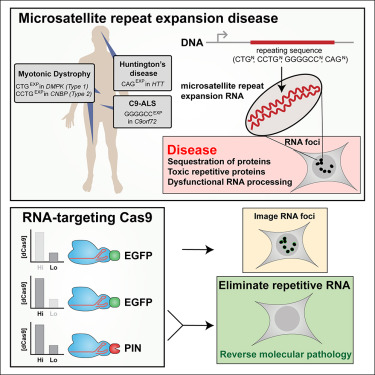
Microsatellite repeat expansion diseases arise because there are errant repeats in RNA sequences that are toxic to the cell, in part because they prevent production of crucial proteins. These repetitive RNAs accumulate in the nucleus or cytoplasm of cells, forming dense knots, called foci.

In this proof-of-concept study, Yeo’s team used RCas9 to eliminate the problem-causing RNAs associated with microsatellite repeat expansion diseases in patient-derived cells and cellular models of the diseases in the laboratory.
Normally, CRISPR-Cas9 works like this: researchers design a “guide” RNA to match the sequence of a specific target gene. The RNA directs the Cas9 enzyme to the desired spot in the genome, where it cuts DNA. The cell repairs the DNA break imprecisely, thus inactivating the gene, or researchers replace the section adjacent to the cut with a corrected version of the gene. RCas9 works similarly but the guide RNA directs Cas9 to an RNA molecule instead of DNA.
The researchers tested the new RCas9 system on microsatellite repeat expansion disease RNAs in the laboratory. RCas9 eliminated 95 percent or more of the RNA foci linked to myotonic dystrophy type 1 and type 2, one type of ALS and Huntington’s disease. The approach also eliminated 95 percent of the aberrant repeat RNAs in myotonic dystrophy patient cells cultured in the laboratory.
Another measure of success centered on MBNL1, a protein that normally binds RNA, but is sequestered away from hundreds of its natural RNA targets by the RNA foci in myotonic dystrophy type 1. When the researchers applied RCas9, they reversed 93 percent of these dysfunctional RNA targets in patient muscle cells, and the cells ultimately resembled healthy control cells.
While this study provides the initial evidence that the approach works in the laboratory, there is a long way to go before RCas9 could be tested in patients, Yeo explained.
One bottleneck is efficient delivery of RCas9 to patient cells. Non-infectious adeno-associated viruses are commonly used in gene therapy, but they are too small to hold Cas9 to target DNA. Yeo’s team made a smaller version of Cas9 by deleting regions of the protein that were necessary for DNA cleavage, but dispensable for binding RNA.
“The main thing we don’t know yet is whether or not the viral vectors that deliver RCas9 to cells would elicit an immune response,” he said. “Before this could be tested in humans, we would need to test it in animal models, determine potential toxicities and evaluate long-term exposure.”
To do this, Yeo and colleagues launched a spin-out company called Locana to handle the preclinical steps required for moving RCas9 from the lab to the clinic for RNA-based diseases, such as those that arise from microsatellite repeat expansions.
“We are really excited about this work because we not only defined a new potential therapeutic mechanism for CRISPR-Cas9, we demonstrated how it could be used to treat an entire class of conditions for which there are no successful treatment options,” said David Nelles, PhD, co-first author of the study with Ranjan Batra, PhD, both postdoctoral researchers in Yeo’s lab.
“There are more than 20 genetic diseases caused by microsatellite expansions in different places in the genome,” Batra said. “Our ability to program the RCas9 system to target different repeats, combined with low risk of off-target effects, is its major strength.”
Cell – Elimination of Toxic Microsatellite Repeat Expansion RNA by RNA-Targeting Cas9
Highlights
•RNA-targeting Cas9 (RCas9) supports efficient targeting of repetitive RNAs
•An RNA endonuclease fused to nuclease-null Cas9 enables an RNA-specific CRISPR system
•An RCas9 system with truncated Cas9 can be packaged in adeno-associated virus
•RCas9 reverses splicing defects in myotonic dystrophy type 1 patient cells
Summary
Microsatellite repeat expansions in DNA produce pathogenic RNA species that cause dominantly inherited diseases such as myotonic dystrophy type 1 and 2 (DM1/2), Huntington’s disease, and C9orf72-linked amyotrophic lateral sclerosis (C9-ALS). Means to target these repetitive RNAs are required for diagnostic and therapeutic purposes. Here, we describe the development of a programmable CRISPR system capable of specifically visualizing and eliminating these toxic RNAs. We observe specific targeting and efficient elimination of microsatellite repeat expansion RNAs both when exogenously expressed and in patient cells. Importantly, RNA-targeting Cas9 (RCas9) reverses hallmark features of disease including elimination of RNA foci among all conditions studied (DM1, DM2, C9-ALS, polyglutamine diseases), reduction of polyglutamine protein products, relocalization of repeat-bound proteins to resemble healthy controls, and efficient reversal of DM1-associated splicing abnormalities in patient myotubes. Finally, we report a truncated RCas9 system compatible with adeno-associated viral packaging. This effort highlights the potential of RCas9 for human therapeutics.

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.