Electrically controlled acid mining of rare earth metals.
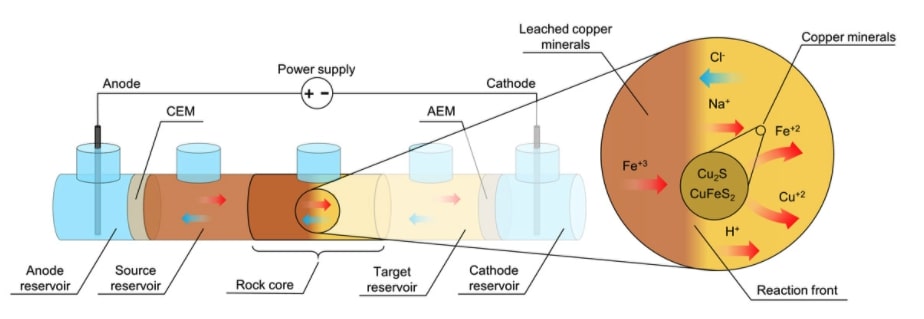
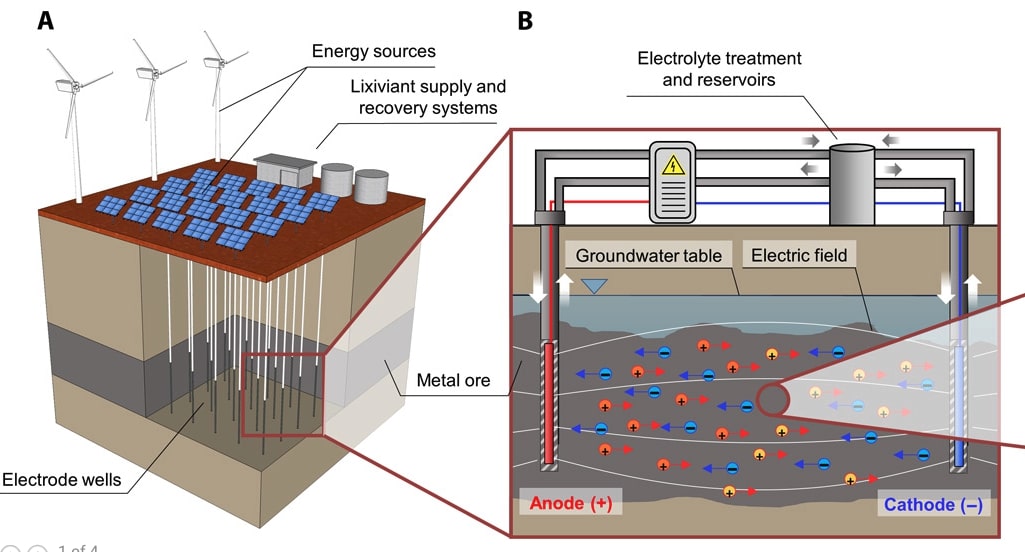
There is a proof of concept to use an electric field to control the movement of an acid within a low permeability copper-bearing ore deposit to selectively dissolve and recover the metal in situ.
This can transform the mining industry because it has the capability to dissolve metals from a wide range of ore deposits that were previously considered inaccessible.
Furthermore, due to the non-invasive nature of the extraction, the research team are hopeful that the study will help usher in a more sustainable future for the industry.
This is like a human keyhole surgery analogy to mining.

Science – Toward a more sustainable mining future with electrokinetic in situ leaching
Abstract
Metals are currently almost exclusively extracted from their ore via physical excavation. This energy-intensive process dictates that metal mining remains among the foremost CO2 emitters and mine waste is the single largest waste form by mass. We propose a new approach, electrokinetic in situ leaching (EK-ISL), and demonstrate its applicability for a Cu-bearing sulfidic porphyry ore. In laboratory-scale experiments, Cu recovery was rapid (up to 57 weight % after 94 days) despite low ore hydraulic conductivity (permeability = 6.1 mD; porosity = 10.6%). Multiphysics numerical model simulations confirm the feasibility of EK-ISL at the field scale. This new approach to mining is therefore poised to spearhead a new paradigm of metal recovery from currently inaccessible ore bodies with a markedly reduced environmental footprint.

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.

Maybe good for asteroids? Bag bola and slag. Extrude mass as cables to charge and transport…e-sail battery
How do you keep the acid from entering the ground water? This is a variation of uranium extraction in the Dakotas, it released uranium into the drinking water.
I'll wait for a real world test. It would be cool if mining costs were reduced though.
Having worked in heap leaching mineral extraction, I would say that maybe 50% of my job involved making sure that none of the leachate could escape into the environment.
The idea that we could just pump it directly into the water table would be… courageous.
Of course a critical factor is what chemicals are in the fluid being used. Both before and after the huge electrochemical reaction. For example, in this case there would be a lot of dissolved copper, which is NOT going to fly in all sorts of places because copper ions are straight out toxic.
What's the environmental impact of the acid?
Can the acid reagent be recovered afterwards? Otherwise, I do not see the greens favouring this over regular digging. Eventually the acid would contaminate the nearby water table unless the mining site were somehow hydrologically insulated?
The article describes a copper ore experiment, with only a single mention of rare earths and other elements.
It looks like an electrically driven in situ heap leach. I couldn't derive the energy efficiency of the process, where is GoatGuy when we need him?
Kurt's comment on asteroid mining neglects the water dependence of the process described. There would also be some temperature effects in space, affecting the efficiency of the ionic migration, as well as the reagent HCl requirements.
This would reduce labor as well. This technique ought to work for asteroid mining as well.