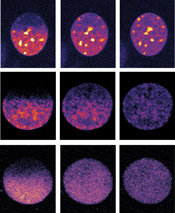
Micropilot detected cells at particular stages of cell division (each row shows one cell’s nucleus), and then instructed the microscope to remove fluorescent tags from proteins in half the cell’s nucleus (left), and record what happened next (middle and right). Image & video credits: EMBL.
The sight of a researcher sitting at a microscope for hours, painstakingly searching for the right cells, may soon be a thing of the past, thanks to new software created by scientists at the European Molecular Biology Laboratory (EMBL) in Heidelberg, Germany. Presented today in Nature Methods, the novel computer programme can rapidly learn what the scientist is looking for and then takes over this laborious and time-consuming task, automatically performing complex microscopy experiments when it detects cells with interesting features.
Called Micropilot, the software brings machine learning to microscopy. It analyses low-resolution images taken by a microscope and, once it has identified a cell or structure the scientists are interested in, it automatically instructs the microscope to start the experiment. This can be as simple as recording high-resolution time-lapse videos or as complex as using lasers to interfere with fluorescently tagged proteins and recording the results.
The software is a boon to systems biology studies, as it generates more data, faster. In a mere four nights of unattended microscope operation, Micropilot detected 232 cells in two particular stages of cell division and performed a complex imaging experiment on them, whereas an experienced microscopist would have to work full-time for at least a month just to find those cells among the many thousands in the sample. With such high throughput, Micropilot can easily and quickly generate enough data to obtain statistically reliable results, allowing scientists to probe the role of hundreds of different proteins in a particular biological process.
Jan Ellenberg and Rainer Pepperkok, whose teams at EMBL designed Micropilot, have used the software to deploy several different microscopy experiments, investigating various aspects of cell division. They determined when structures known as endoplasmic reticulum exit sites form, and uncovered the roles of two proteins, CBX1 and CENP-E, in condensing genetic material into tightly-wound chromosomes and in forming the spindle which helps align those chromosomes. This software will be a key tool for the European systems biology projects Mitosys and SystemsMicroscopy, for which Ellenberg and Pepperkok are developing technology.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.