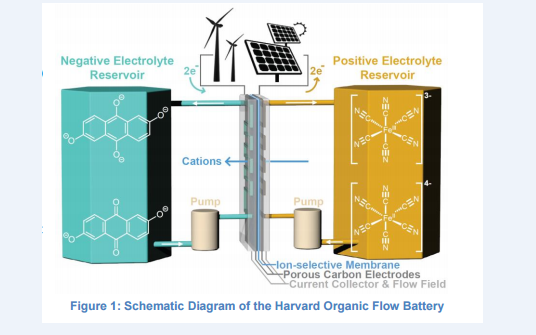
A Harvard team is addressing the challenges of grid storage by designing a flow battery based on inexpensive organic molecules in aqueous (water-based) electrolyte. The team has focused on non-toxic quinone molecules, which can be found in plants such as rhubarb, as an electroactive chemical that can reversibly store energy in a water-based solution at room temperature. The group employed theoretical and organic synthetic methods to evaluate hundreds of thousands of possible quinone-based chemicals that might offer the necessary electrochemical potential, solubility in water, and thermodynamic stability. The first demonstration of these systems, 2,7-anthraquinone disulfonic acid coupled to a bromine solution, has a reduction-oxidation window of 0.8 V. Details of this early, proof of concept battery were published in 2014
Professor Michael Aziz, leader of the Harvard project, said there are still problems to sort out with the “calendar life” of storage chemicals but the basic design is essentially proven.
“We have a fighting chance of bringing down the capital cost to $100 a kilowatt hour, and that will change the world. It could complement wind and solar on a very large scale,” he told the Daily Telegraph.
The latest refinement is to replace toxic bromine with harmless ferrocyanide – used in food additives. The battery cannot catch fire. It is safe and clean. “This is chemistry I’d be happy to put in my basement,” he said.
The design is delightfully simple. It uses a tank of water. You could have one at home in Los Angeles, Lagos, Buenos Aires, Delhi, or Guangzhou, storing solar power in the day to drive your air-conditioning at night. It could be scaled up for a 500 megawatt wind farm.
Italy’s Green Energy Storage has the European licence. It is building a prototype with the Kessler Foundation at Trento University, backed by EU funds. “We have a chemistry that is ten times cheaper than anything on the market,” said Salvatore Pinto, the chairman.
“We are speaking to three utilities in Europe and we will install our first battery as a field test next year,” he said.
It is a race. Tim Grejtak, an energy expert at Lux Research, said Lockheed Martin and Pacific Northwest labs are both working on their own organic flow batteries, while others are developing variants with designed molecules.
Since these experiments were completed, the group has moved to make their system cheaper and less toxic, and to increase the voltage to 1.2 V. To date, the team has analyzed over one million quinone-based molecules using computer simulation, with thousands of them determined to be promising for storage. Of these, a dozen were selected for synthesis and testing, based upon their expected synthetic feasibility, electrochemical redox potential, aqueous solubility, and chemical stability during operation.
The team has developed a prototype cell design using safer alkaline liquids, no precious metals, and other battery components made of plastic or inexpensive metals with coatings proven to protect against corrosion. The team is advancing this innovative storage concept with goals of improving performance, including power density, cycle life and round trip efficiency. Their goal is to have a pilot-sale system ready for external testing by the end of the performance period.
Pathway to Economic Impact
Sustainable Innovations, Harvard’s commercialization partner, is developing a 3 kW system incorporating Harvard’s chemistry for demonstration by the end of the performance period. Sustainable Innovations is currently raising funding to complete
commercialization of a low-cost electrochemical platform that will support the launch of a series of products that meet critical market needs. If Harvard’s innovative storage concept proves commercially competitive, this technology can be inserted into a low-cost platform to yield a range of quinone based battery systems, with toxicity, cost and performance features that suit a variety of applications ranging from uninterruptable power supplies through micro-grid back-up, to full grid support.
Technoeconomic analysis and customer interest will determine the top candidates resulting from this project. The relatively non-toxic chemicals used in the battery design are widely used in the textile dye industry. Because these materials are already in commercial production, it is likely that scale-up for the organic battery will be rapid and cost effective.
Long Term Impacts
The use of redox-active organics in flow batteries has the potential to significantly drop the cost and toxicity of large scale grid storage batteries. This specific concept of using water-soluble quinones is unprecedented for flow batteries, and now provides a proof-of-concept for low-cost, more environmentally benign alternatives to the accepted chemistries of the past. With continuing development, and scale up to manufacturing in large quantities, redox-active organic battery technology has the potential for a highly competitive evolution of costs
Background Research Papers
B. Huskinson, M.P. Marshak, C. Suh, S. Er, M.R. Gerhardt, C.J. Galvin, X. Chen, A. Aspuru-Guzik, R.G. Gordon and M.J. Aziz, “A metal-free organic-inorganic aqueous flow battery”, Nature 505, 195 (2014).
Kaixiang Lin, Qing Chen, Michael R. Gerhardt, Liuchuan Tong, Sang Bok Kim, Louise Eisenach, Alvaro W. Valle, David Hardee, Roy G. Gordon, Michael J. Aziz, and Michael P. Marshak, Alkaline Quinone Flow Battery, Science 349, 1529 (2015). Qing Chen, Louise Eisenach, and Michael J. Aziz, Cycling analysis of a quinone-bromide redox flow battery, Journal of The Electrochemical Society 163:1, A5057-A5063 (2016).
Suleyman Er, Changwon Suh, Michael Marshak, Alan Aspuru-Guzik, “Computational design of molecules for an all-quinone redox flow battery”, Chemical Sciences, 6, 2015, 885-893.
Qing Chen, Michael R. Gerhardt, Lauren Hartle and Michael J. Aziz, “A Quinone-Bromide Flow Battery with 1 W/cm2 Power Density”, Journal of the Electrochemical Society, Vol 163: 1, 2016, A5010-A5013.
Brian Huskinson, Michael P. Marshak, Michael R. Gerhardt, Michael J. Aziz “Cycling of a Quinone-Bromide Flow Battery for Large Scale Electrochemical Energy Storage”, ECS Transactions, 61, 2014.
SOURCES- ARPA-E, Harvard, Telegraph UK

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.