Ads : Nano Technology Netbook Technology News Computer Software
A new technology developed at Hadassah University Medical Center has made it possible to produce large amounts of human embryonic stem cells for industry and research by growing them in suspension. Instead of being grown on beds in one layer, the embryonic stem cells could be cultivated in large vats with precise, computerized monitoring of growth conditions.
Until now, hESCs – which have the potential of ripening into any other type of body cells for repair of tissues and organs – have been created in small numbers.
Called a “breakthrough” in hESC technology, the research was just published in the prestigious science journal Nature Biotechnology. The article, titled “Derivation, propagation and controlled differentiation of human embryonic stem cells in suspension,” was written by Dr. Debora Steiner as part of her post-doctoral work under the direction of Prof. Benjamin Reubinoff, both of Hadassah’s Human Embryonic Stem Cell Research Center, with participation by other Hadassah colleagues.
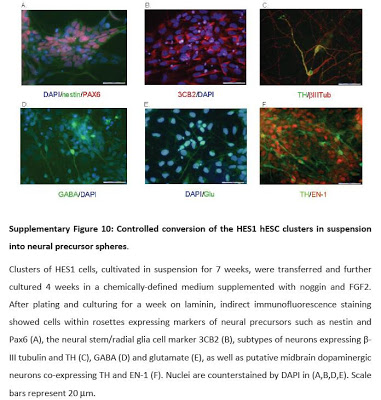
Undifferentiated human embryonic stem cells (hESCs) are currently propagated on a relatively small scale as monolayer colonies. Culture of hESCs as floating aggregates is widely used for induction of differentiation into embryoid bodies8. Here we show that hESC lines can be derived from floating inner cell masses in suspension culture conditions that do not involve feeder cells or microcarriers. This culture system supports prolonged propagation of the pluripotent stem cells as floating clusters without their differentiation into embryoid bodies. HESCs cultivated as aggregates in suspension maintain the expression of pluripotency markers and can differentiate into progeny of the three germ layers both in vitro and in vivo. We further show the controlled differentiation of hESC clusters in suspension into neural spheres. These results pave the way for large-scale expansion and controlled differentiation of hESCs in suspension, which would be valuable in basic and applied research.
10 pages of supplemental information
It was thought that propagation of hESCs in a suspension (a mixture in which fine particles are suspended in a fluid where they are supported by buoyancy) was impossible because under these conditions, the cells would stop multiplying and instead undergo uncontrolled ripening into various types of cells.
But the Hadassah researchers developed unique conditions that made it possible for the cells to multiply significantly in suspension without differentiating into specific cell types. By changing the conditions of the culture, they showed that they could control the ripening of hESC clusters in suspension into neural spheres. Thus instead of being grown on beds in one layer, the stem cells could be cultivated in large vats with precise, computerized monitoring of growth conditions.
Other Stem Cell Research
Researchers believe that umbilical cord blood could serve as a universal source of stem cells for all patients who need a stem cell transplant, but the numbers of stem cells in cord blood units are limited, so there is a clinical need to develop a method to expand cord blood stem cells for transplantation purposes. “Unfortunately, there are no soluble growth factors identified to date that have been proven to expand human stem cells for therapeutic purposes,” said John Chute, M.D., a stem cell transplant physician and cell biologist at Duke and senior author of the paper.
Chute, working with Heather Himburg, a post-doctoral fellow in his laboratory, discovered that adding pleiotrophin, a naturally-occurring growth factor, stimulated a ten-fold expansion of stem cells taken from the bone marrow of a mouse.
They also found that pleiotrophin increased the numbers of human cord blood stem cells in culture that were capable of engraftment in immune-deficient mice. When they injected pleiotrophin into mice that had received bone marrow-suppressive radiation, they observed a 10-fold increase in bone marrow stem cells compared to untreated mice. “These results confirmed that pleiotrophin induces stem cell regeneration following injury,” said Chute.
Chute says the finding could lead to broader application of cord blood transplants for the large numbers of patients who do not have an immune-matched donor “Perhaps more importantly, systemic treatment with pleiotrophin may have the potential to accelerate recovery of the blood and immune system in patients undergoing chemotherapy or radiotherapy,” he said.
Advertising
Trading Futures
Nano Technology
Netbook Technology News
Computer Software
Future Predictions
Thank You

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.