A new cancer drugs work by targeting tumors by their genetic make-up instead of where originate in the body.

On November 26, 2018, the Food and Drug Administration granted accelerated approval to larotrectinib (VITRAKVI, Loxo Oncology Inc. and Bayer) for adult and pediatric patients with solid tumors that have a neurotrophic receptor tyrosine kinase (NTRK) gene fusion without a known acquired resistance mutation, that are either metastatic or where surgical resection is likely to result in severe morbidity, and who have no satisfactory alternative treatments or whose cancer has progressed following treatment. It was the second tissue-agnostic FDA approval for the treatment of cancer.
In 2017, FDA granted its first tissue-agnostic approval (pembrolizumab for patients with unresectable or metastatic, microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors) and first tissue-agnostic orphan-drug designations (larotrectinib and entrectinib, each for the treatment of solid tumors with NTRK-fusion proteins).
Recent developments in the genomics of oncogenesis have opened the possibility of a molecular
marker defining a disease that spans multiple histology-based tumors in a tissue agnostic manner. However, it is also clear that tumors represent heterogeneous disease states and that histologic context may be of importance in evaluating potential targeted treatments.
They are able to shrink tumors in up to three-quarters of cancers tested. Preparations are underway to ensure the next generation of treatment can be quickly made available to patients. Two of the first drugs are expected to be licensed later this year, and could be approved by NHS (UK National Health) rationing bodies soon after, depending on price negotiations.
Two drugs – Larotrectinib, produced by Bayer, and entrectinib, from Roche, are expected to be the first drugs to be licensed in the UK later in 2019.
Around 850 patients a year in the UK could benefit from the first genetic targeting drugs while many thousands a year in the UK are eventually expected to benefit from other treatments on the horizon.
The drugs work by blocking the NTRK enzyme, effectively shrinking the tumor. Early clinical trials showed the tumor responded in two thirds to three-quarters of the cancers tested.
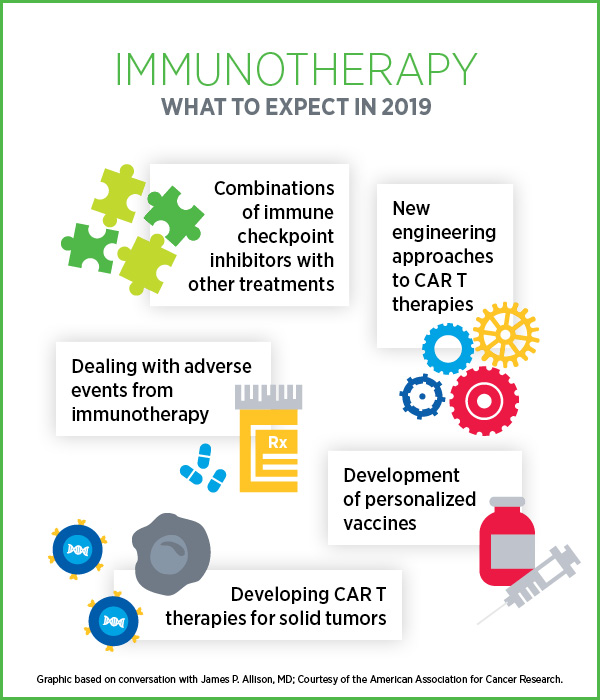
Personalized Cancer Vaccines and New CAR-T Approvals

SOURCES – AACR – American Association for Cancer Research, FDA, Telegraph UK
Written By Brian Wang, Nextbigfuture.com

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.