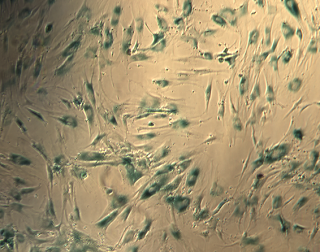
These blue cells are human mesenchymal stem cells that have become senescent –- or lost the ability to divide — after X-ray irradiation. This image, obtained using senescence-associated β-gal staining, helped Berkeley Lab scientists better understand the process that triggers senescence in mesenchymal stem cells.
Their research can be used to help refine cancer treatments that utilize ionizing radiation, and may help inform future work to protect the health of astronauts on missions to deep space. It also sheds light on cellular senescence, a process in which cells permanently stop dividing that is linked to cancer and aging.
Lab scientists zeroed in on the protein that triggers senescence in human mesenchymal stem cells. Found in the bone marrow of juveniles and adults, these stem cells are critical for maintaining and repairing tissues such as bone, cartilage, and muscle.Like all stem cells, human mesenchymal stem cells have the ability to proliferate and differentiate into specialized cell types. They sometimes stop dividing when they’re damaged, however, which can contribute to the transformation of normal cells into cancer cells and is believed to play a role in the aging process.
Now scientists have a better understanding of how this breakdown occurs.
“We found that X-ray induced cellular senescence of human mesenchymal stem cells is a highly complex process that is mediated by a critical protein kinase called CK2,” says Daojing Wang of Berkeley Lab’s Life Sciences Division, the principal investigator of a study that is published in the October 15, 2009 issue of the journal Cancer Research.
Separate Stem Cell Research Funding
The UCLA disease team grants were awarded to:
Irvin Chen
Chen, director of the UCLA AIDS Institute and a professor of microbiology, immunology and molecular genetics, received $19,999,580 to develop a method to block HIV infection and reproduction in the human body.Dr. Donald Kohn
Kohn, director of the Human Gene Medicine Program at UCLA and a professor of microbiology, immunology and molecular genetics and of pediatrics, received $9,212,365 to develop a blood stem cell transplant to cure sickle cell disease.Dr. Dennis Slamon
Slamon, director of clinical/translational research at UCLA’s Jonsson Comprehensive Cancer Center and chief of the division of hematology and oncology at UCLA, received $19,979,660 to develop drugs that target cancer stem cells, believed to be the cause of some cancers, including glioblastoma (brain), ovarian and colorectal cancers

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.