A team of researchers with the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) has invented a technique in which DNA or RNA assays — the key to genetic profiling and disease detection — can be read and evaluated without the need of elaborate chemical labeling or sophisticated instrumentation.
Based on electrostatic repulsion — in which objects with the same electrical charge repel one another — the technique is relatively simple and inexpensive to implement, and can be carried out in a matter of minutes.
“One of the most amazing things about our electrostatic detection method is that it requires nothing more than the naked eye to read out results that currently require chemical labeling and confocal laser scanners,” said Jay Groves, a chemist with joint appointments at Berkeley Lab’s Physical Biosciences Division and the Chemistry Department of the University of California (UC) at Berkeley, who led this research. “We believe this technique could revolutionize the use of DNA microarrays for both research and diagnostics.”
Groves, who is also a Howard Hughes Medical Institute (HHMI) investigator, and members of his research group Nathan Clack and Khalid Salaita, have published a paper on their technique in the journal Nature Biotechnology, which is now available online. The paper is entitled “Electrostatic readout of DNA microarrays with charged microspheres.”
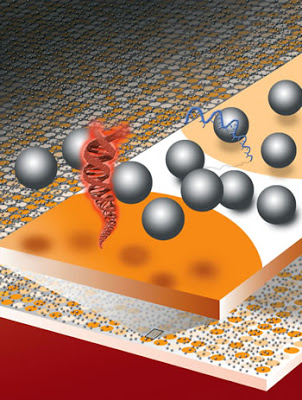
In their paper, Groves, Clack, and Salaita describe how dispersing a fluid containing thousands of electrically-charged microscopic beads or spheres made of silica (glass) across the surface of a DNA microarray and then observing the Brownian motion of the spheres provides measurements of the electrical charges of the DNA molecules. These measurements can in turn be used to interrogate millions of DNA sequences at a time. What’s more, these measurements can be observed and recorded with a simple hand-held imaging device — even a cell phone camera will do.
“The assumption has been that no detection technique could be more sensitive than fluorescent labeling, but this is completely untrue, as our results have plainly demonstrated,” said Groves. “We’ve shown that changes in surface charge density as a result of specific DNA hybridization can be detected and quantified with 50-picometer sensitivity, single base-pair mismatch selectivity, and in the presence of complex backgrounds. Furthermore, our electrostatic detection technique should render DNA and RNA microarrays sufficiently cost effective for broad world-health applications, as well as research.”
FURTHER READING
Nature biotechnology paper: Electrostatic readout of DNA microarrays with charged microspheres
DNA microarrays are used for gene-expression profiling, single-nucleotide polymorphism detection and disease diagnosis1, 2, 3. A persistent challenge in this area is the lack of microarray screening technology suitable for integration into routine clinical care4, 5. Here, we describe a method for sensitive and label-free electrostatic readout of DNA or RNA hybridization on microarrays. The electrostatic properties of the microarray are measured from the position and motion of charged microspheres randomly dispersed over the surface. We demonstrate nondestructive electrostatic imaging with 10-m lateral resolution over centimeter-length scales, which is four-orders of magnitude larger than that achievable with conventional scanning electrostatic force microscopy. Changes in surface charge density as a result of specific hybridization can be detected and quantified with 50-pM sensitivity, single base-pair mismatch selectivity and in the presence of complex background. Because the naked eye is sufficient to read out hybridization, this approach may facilitate broad application of multiplexed assays

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.