In a study published July 11 in the peer-reviewed Journal of Virology, researchers from the UCLA AIDS Institute and Center for AIDS Research found that recently discovered potent antibodies can be used to generate a specific type of cell called chimeric antigen receptors, or CARs, that can be used to kill cells infected with HIV-1.
CARs are artificially created immune T cells that have been engineered to produce receptors on their surface that are designed to target and kill specific cells containing viruses or tumor proteins. Chimeric receptors are the focus of ongoing research into how gene immunotherapy can be used to fight cancer. But they could also be used to create a strong immune response against HIV, said Dr. Otto Yang, professor of medicine in the division of infectious diseases at the David Geffen School of Medicine at UCLA and the study’s corresponding author.
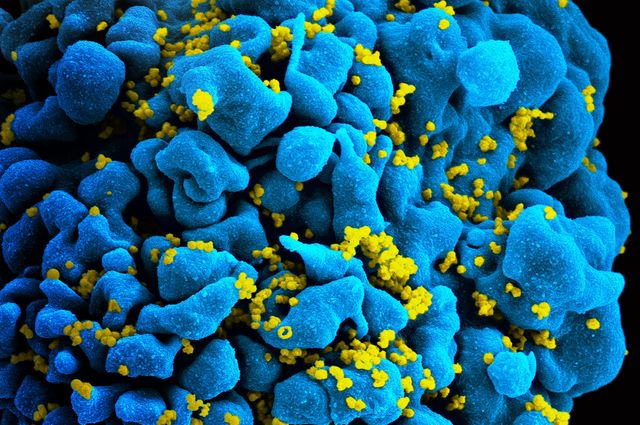
Researchers find that potent antibodies can be used to generate a specific type of cell that can be used to kill cells infected with HIV-1. An HIV-infected T cell is shown here.
Journal of Virology – HIV-1-Specific Chimeric Antigen Receptors Based on Broadly Neutralizing Antibodies
Although the human body’s immune system does initially respond to and attack HIV, the sheer onslaught of the virus — its ability to hide in different T cells and to rapidly replicate — eventually wears out and destroys the immune system, leaving the body vulnerable to a host of infections and diseases. Researchers have been looking for ways to strengthen the immune system against HIV, and it now appears CARs could be a weapon in that fight.
“We took new generation antibodies and engineered them as artificial T-cell receptors, to reprogram killer T cells to kill HIV-infected cells,” said Yang, who is also director of vaccine and pathogenesis research at the AIDS Institute and Center for AIDS Research. “Others have used antibodies against cancer antigens to make artificial T-cell receptors against cancer and shown this to be helpful in cancer treatment.” UCLA is the first to design this strategy for HIV.
While the receptors approach has been in use for almost 10 years to fight cancer, this is the first attempt to use the technique to treat HIV since 15 years ago, when experiments proved unsuccessful. The new research differs because it takes advantage of new antibodies that have been discovered in the past few years. In the previous trials, researchers had used an early type that was not antibody-based. That approach, however, was abandoned because it was clinically ineffective.
Here the researchers used seven recently discovered “broadly neutralizing antibodies” that have the ability to bind multiple strains of invading viruses, unlike earlier isolated antibodies that tend to bind few strains. These antibodies were re-engineered as artificial CAR-T cell receptors to have activity against broad strains of HIV. In lab tests, the researchers found that all seven had varying degrees of ability to direct killer T cells to proliferate, kill and suppress viral replication in response to HIV-infected cells.
Yang notes that “what works in a test tube doesn’t necessarily work in a person,” so the next step is to find strategies to put these receptors into humans. But this therapy shows enough promise to move forward with further research.
Abstract
Although the use of chimeric antigen receptors (CARs) based on single-chain antibodies for gene immunotherapy of cancers is increasing due to promising recent results, the earliest CAR therapeutic trials were done for HIV-1 infection in the late 1990s. This approach utilized a CAR based on human CD4 as a binding domain and was abandoned for a lack of efficacy. The growing number of HIV-1 broadly neutralizing antibodies (BNAbs) offers the opportunity to generate novel CARs that may be more active and revisit this modality for HIV-1 immunotherapy. We used sequences from seven well-defined BNAbs varying in binding sites and generated single-chain-antibody-based CARs. These CARs included 10E8, 3BNC117, PG9, PGT126, PGT128, VRC01, and X5. Each novel CAR exhibited conformationally relevant expression on the surface of transduced cells, mediated specific proliferation and killing in response to HIV-1-infected cells, and conferred potent antiviral activity (reduction of viral replication in log10 units) to transduced CD8+ T lymphocytes. The antiviral activity of these CARs was reproducible but varied according to the strain of virus. These findings indicated that BNAbs are excellent candidates for developing novel CARs to consider for the immunotherapeutic treatment of HIV-1.
Importance
While chimeric antigen receptors (CARs) using single-chain antibodies as binding domains are growing in popularity for gene immunotherapy of cancers, the earliest human trials of CARs were done for HIV-1 infection. However, those trials failed, and the approach was abandoned for HIV-1. The only tested CAR against HIV-1 was based on the use of CD4 as the binding domain. The growing availability of HIV-1 broadly neutralizing antibodies (BNAbs) affords the opportunity to revisit gene immunotherapy for HIV-1 using novel CARs based on single-chain antibodies. Here we construct and test a panel of seven novel CARs based on diverse BNAb types and show that all these CARs are functional against HIV-1.
SOURCES- UCLA, Journal of Virology

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.