Ad Support : Nano Technology Netbook Technology News Computer Software
Breakthrough in cell regeneration could lead to the repair of damaged hearts. Scientists have tried for 20 years to convert nonmuscle cells into heart muscle, but it turns out we just needed the right combination of genes at the right dose.
Cell Journal – Direct Reprogramming of Fibroblasts into Functional Cardiomyocytes by Defined Factors
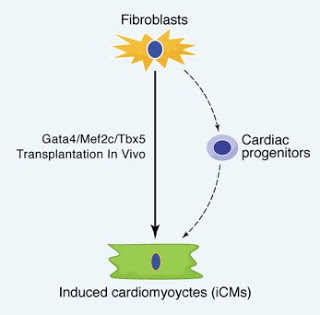
The reprogramming of fibroblasts to induced pluripotent stem cells (iPSCs) raises the possibility that a somatic cell could be reprogrammed to an alternative differentiated fate without first becoming a stem/progenitor cell. A large pool of fibroblasts exists in the postnatal heart, yet no single “master regulator” of direct cardiac reprogramming has been identified. Here, we report that a combination of three developmental transcription factors (i.e., Gata4, Mef2c, and Tbx5) rapidly and efficiently reprogrammed postnatal cardiac or dermal fibroblasts directly into differentiated cardiomyocyte-like cells. Induced cardiomyocytes expressed cardiac-specific markers, had a global gene expression profile similar to cardiomyocytes, and contracted spontaneously. Fibroblasts transplanted into mouse hearts one day after transduction of the three factors also differentiated into cardiomyocyte-like cells. We believe these findings demonstrate that functional cardiomyocytes can be directly reprogrammed from differentiated somatic cells by defined factors. Reprogramming of endogenous or explanted fibroblasts might provide a source of cardiomyocytes for regenerative approaches.
Heart disease is a leading cause of adult and childhood mortality. The underlying pathology is typically loss of cardiomyocytes that leads to heart failure or improper development of cardiomyocytes during embryogenesis that leads to congenital heart malformations. Because postnatal cardiomyocytes have little or no regenerative capacity, current therapeutic approaches are limited. Embryonic stem cells possess clear cardiogenic potential, but efficiency of cardiac differentiation, risk of tumor formation, and issues of cellular rejection must be overcome.
In this study, we examined whether key developmental cardiac regulators could reprogram cardiac fibroblasts into cardiomyocytes. We found that out of a total of 14 factors, a specific combination of three transcription factors, Gata4, Mef2c, and Tbx5, was sufficient to generate functional beating cardiomyocytes directly from mouse postnatal cardiac or dermal fibroblasts and that the induced cardiomyocytes (iCMs) were globally reprogrammed to adopt a cardiomyocyte-like gene expression profile.
Here we demonstrated that the combination of three transcription factors, Gata4, Mef2c, and Tbx5, can rapidly and efficiently induce cardiomyocyte-like cells from postnatal cardiac and dermal fibroblasts. iCMs were similar to neonatal cardiomyocytes in global gene expression profile, electrophysiologically, and could contract spontaneously, demonstrating that functional cardiomyocytes can be generated from differentiated somatic cells by defined factors. Although much refinement and characterization of the reprogramming process will be necessary, the findings reported here raise the possibility of reprogramming the vast pool of endogenous fibroblasts that normally exists in the heart into functional cardiomyocytes for regenerative purposes.Several lines of evidence suggest that the iCMs we describe here originated from differentiated fibroblasts. We found that any potential rare cardiac progenitor-like cells, marked by c-kit or Isl1, were dispensable for cardiomyocyte induction (Beltrami et al., 2003). Furthermore, the high efficiency of cardiac induction (up to 20%) does not favor the interpretation that rare stem or progenitor cells were the origin of induced cardiomyocytes. Most importantly, the ability to reprogram dermal fibroblasts into iCMs supports the conclusion that cardiac progenitors are not the target cells for the reprogramming factors. Remarkably, reprogramming of cardiac fibroblasts to myocytes occurred in a relatively short period, with the first GFP+ cells appearing at day 3, in contrast to iPSC reprogramming, which typically takes 1020 days and occurs with much lower efficiency (< 0.1%) (Takahashi and Yamanaka, 2006). Despite the early initiation of reprogramming, the process appears to continue for several weeks, with progressive changes in gene expression, contractile ability, and electrophysiologic maturation. Although many questions remain regarding the mechanisms of reprogramming, we were able to genetically test the route of cell fate alteration. Our findings suggest that cardiomyocytes were directly induced from cardiac fibroblasts without reverting to a cardiac progenitor cell state, which may explain the rapid early reprogramming process. This conclusion was supported by the absence of Isl1-Cre-YFP or Mesp1-Cre-YFP activation during the process of reprogramming, which would have marked any cells that transiently expressed Isl1 or Mesp1
The ability to reprogram endogenous cardiac fibroblasts into cardiomyocytes has many therapeutic implications. First, the avoidance of reprogramming to pluripotent cells before cardiac differentiation would greatly lower the risk of tumor formation in the setting of future cell-based therapies. Second, large amounts of an individual’s own fibroblasts can be grown from a cardiac biopsy or skin biopsy in vitro for transduction with the defined factors, followed by delivery of cells to damaged hearts. Third, and most promising, is the potential to introduce the defined factors, or factors that mimic their effects, directly into the heart to reprogram the endogenous fibroblast population, which represents more than 50% of the cells, into new cardiomyocytes that can contribute to the overall contractility of the heart. Our observation that injection of fibroblasts into the heart only 1 day after induction of Gata4/Mef2c/Tbx5 resulted in reprogramming of the transplanted cells suggests that this may be possible. Future studies in human cells and advances in safe delivery of defined factors will be necessary to advance this technology for potential regenerative therapies.
If you liked this article, please give it a quick review on Reddit, or StumbleUpon. Thanks
Supporting Advertising
Business Success
How to Make Money
Executive Jobs
Paid Surveys
Thank You

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.