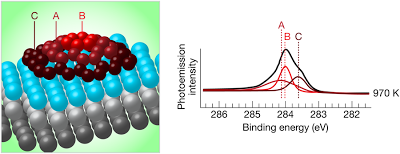
Left) Carbon dome structure on an iridium substrate, indicating the tightly bound atoms at the edge of the island (C), the weakly bound atoms at the center (B) and intermediate atoms (A). (Right) Photoelectron spectrum at 970 K showing the contributions from the edge (C), center (B), and intermediate (A) atoms in the dome structure. (In this simplified schematic diagram, the dome structure is shown to consist of three types of carbon atoms; the actual structure is more complex.)
1. Photoelectron spectroscopy data that suggests that en route to forming continuous sheets, graphene islands grow on an iridium surface in the form of microscopic domes. According to their model, the domes consist of circular islands of graphene that are attached via strong chemical bonds to the close-packed iridium surface at the island’s perimeter, but are not chemically attached in the center. The islands grow by attaching atoms and smaller islands to their edges.
Harnessing the potential of graphene in electronic devices requires a method for producing it in sizes larger than available graphite crystals, and surface scientists have been concentrating on developing methods of producing large-scale and perfect graphene films.
The results suggest that the Ir(111) surface catalyzes the growth at the edges of the graphene islands, and that hydrogen does not play any active role in the formation of the graphene films. The picture presented is one where free carbon atoms and small carbon clusters that result from the hydrocarbon decomposition diffuse on the surface to form the islands. It will be very interesting to explore the release of hydrogen as the hydrocarbon molecules react to form graphene. This process would leave islands that are hydrogen terminated without dangling bonds that bind to the metallic surface, producing flat graphene even for small islands. If controlled experiments can produce films having different amounts of hydrogen, then the ability to fabricate films that include both conducting graphene and insulating “graphane”—a two-dimensional layer of covalently bonded hydrocarbon—cannot be far behind. With this ability, the possibilities of producing graphene electronic devices, controlling the work function and optical properties of these materials are endless. Graphene layers might then replace much thicker layers currently used in electronic microcircuits, making future computers and other devices more compact and energy efficient.
2. Fractional quantum Hall effect and insulating phase of Dirac electrons in graphene
New findings, previously considered possible by physicists but only now being seen in the laboratory, show that electrons in graphene can interact strongly with each other. The behavior is similar to superconductivity observed in some metals and complex materials, marked by the flow of electric current with no resistance and other unusual but potentially useful properties. In graphene, this behavior results in a new liquid-like phase of matter consisting of fractionally charged quasi-particles, in which charge is transported with no dissipation.
In graphene, which is an atomic layer of crystalline carbon, two of the distinguishing properties of the material are the charge carriers’ two-dimensional and relativistic character. The first experimental evidence of the two-dimensional nature of graphene came from the observation of a sequence of plateaus in measurements of its transport properties in the presence of an applied magnetic field. These are signatures of the so-called integer quantum Hall effect. However, as a consequence of the relativistic character of the charge carriers, the integer quantum Hall effect observed in graphene is qualitatively different from its semiconductor analogue. As a third distinguishing feature of graphene, it has been conjectured that interactions and correlations should be important in this material, but surprisingly, evidence of collective behaviour in graphene is lacking. In particular, the quintessential collective quantum behaviour in two dimensions, the fractional quantum Hall effect (FQHE), has so far resisted observation in graphene despite intense efforts and theoretical predictions of its existence. Here we report the observation of the FQHE in graphene. Our observations are made possible by using suspended graphene devices probed by two-terminal charge transport measurements. This allows us to isolate the sample from substrate-induced perturbations that usually obscure the effects of interactions in this system and to avoid effects of finite geometry. At low carrier density, we find a field-induced transition to an insulator that competes with the FQHE, allowing its observation only in the highest quality samples. We believe that these results will open the door to the physics of FQHE and other collective behaviour in graphene.
2 pages of Supplemental information
Graphene-gold based DNA sensors will have enhanced sensitivity.

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.