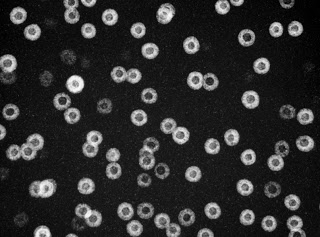
When aluminum is alloyed with the right proportions of scandium and lithium through a simple series of heat treatments, nanoparticle inclusions form in the aluminum matrix (dark background) whose cores, made of aluminum, scandium, and lithium (dark circles), vary in diameter and whose shells, made of aluminum and lithium (bright rings), vary in thickness. But their overall diameters are remarkably uniform.
Scientists have now combined atomic-scale observations with the powerful TEAM microscope at Berkeley Lab’s National Center for Electron Microscopy (NCEM) with atom-probe tomography and other experimental techniques, and with theoretical calculations, to reveal how nanoparticles consisting of cores rich in scandium and surrounded by lithium-rich shells can disperse in remarkably uniform sizes throughout a pure aluminum matrix.
“With the TEAM microscope we were able to study the core-shell structure of these nanoprecipitates and how they form spheres that are nearly the same in diameter,” says Dahmen, the director of NCEM and an author of the Nature Materials paper describing the new studies. “What’s more, these particles don’t change size over time, as most precipitates do. Typically, small particles get smaller and large particles get larger, a process called ripening or coarsening, which eventually weakens the alloys. But these uniform core-shell nanoprecipitates resist change.”
Evolution of an alloy
In the aluminum-scandium-lithium system the researchers found that, after the initial melt, a simple two-step heating process creates first the scandium-rich cores and then the lithium-rich shells of the spherical particles. The spheres self-limit their growth to achieve the same outer dimensions, yielding a lightweight, potentially heat- and corrosion-resistant, superstrong alloy.
“Scandium is the most potent strengthener for aluminum,” says NCEM’s Radmilović, who is also a professor of metallurgy at the University of Belgrade, Serbia, and an author of the Nature Materials paper. “Adding less than one percent scandium can make a dramatic difference in mechanical strength, fracture resistance, corrosion resistance – all kinds of properties.” Because scandium diffuses very slowly through the solid aluminum matrix, the solid mix must be heated to a high temperature (short of melting) before scandium will precipitate.
Lithium is the lightest of all metals (only hydrogen and helium are lighter) and brings not only lightness to an aluminum alloy but, potentially, strength as well. Lithium diffuses much more rapidly than scandium, at much lower temperature.
“The problem is that, by itself, lithium may not live up to its promise,” says Dahmen, a long-time collaborator with Radmilović. “The trick is to convince the lithium to take on a useful crystalline structure, namely L12.”
The L12 unit cell resembles a face-centered cubic cell, among the simplest and most symmetric of crystal structures. Atoms occupy each corner of an imaginary cube and are centered in the cube’s six faces; in the L12 structure, the kinds of atoms at the corners may differ from those at the centers of the faces. For alloy inclusions it’s one of the strongest and stablest of structures because, as Dahmen explains, “once atoms are in place in L12, it’s difficult for them to move.”
Dahmen credits Radmilović with the “intuition” to alloy both scandium and lithium with aluminum, heating and cooling the material in a specific series of steps. That intuition was based on Radmilović’s long experience with the separate properties of aluminum-lithium and aluminum-scandium alloys and a deep understanding of how they were likely to interact. He drew up a recipe for the proportions of ingredients in the initial melt and how to cool and rewarm them.
The key to the process was to use lithium as a kind of catalyst to force a “burst of nucleation” in the scandium. After the three metals are mixed, melted, and quickly cooled or quenched, lithium serves to lower the heating needed to coax scandium to form dense core structures – although the solid mix must still be heated to 450 degrees Celsius (842 Fahrenheit) for 18 hours to form these cores, made of aluminum, lithium, and scandium. The cores average a little over nine nanometers in diameter but are not uniform in size.
Next the alloy is heated again, this time to 190˚ Celsius (374˚ F) for four hours. At the lower temperature the scandium is immobile; the freely-moving lithium forms a shell around the scandium-rich cores, much as water in a cloud crystallizes around a speck of dust to make a snowflake. The shells average about 10.5 nanometers in thickness, but their thickness is not uniform.
What’s remarkable, though, is that when a core is thicker than the average, the shell is thinner than the average, and vice versa: the smaller the core, the faster the shell grows. Core size and shell size are “anticorrelated” and the result is “size focused.” Whole spheres still vary somewhat, but the differences are much less than among the cores alone or the shells alone.
The L12 structure is shown at lower left, with aluminum atoms in gray and scandium or lithium atoms reddish green. In images of a core-shell nanoparticle made by NCEM’s TEAM microscope, each dot shows the top of a column of atoms; the kinds of atoms in each column can be calculated from the brightness and contrast of the dots. The aluminum matrix has a face-centered cubic structure in which all the atoms are aluminum, while in the L12 structure the face-centered positions are also aluminum. But in the core of the nanoparticle (upper right), the columns at the corners of the L12 unit cell are a mix of aluminum, lithium, and scandium atoms, while in the surrounding shell (lower right), the corner columns are a mix of aluminum and lithium
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.