Ad Support : Nano Technology Netbook Technology News Computer Software
* While trying to build computational systems, he is working toward general-purpose manufacturing.
* He called for getting beyond pure self-assembly: You can do some things with passive self-assembly; you can do them much better if you’re able to implement molecular motors.
* self-assembling tiles with carefully designed matching edges can implement any algorithmically-describable shape. And, of course, DNA can implement tiles with lots of different edge configurations.
* He has a slide titled “Theory of Active Self-Assembly (of Molecular Robots).” It includes the statement, “Any algorithmically-describable shape can be assembled _efficiently_.” He talked about shapes with a billion components and said they could be assembled exponentially. He acknowledges “This is something for the future” since we’re still at the stage of molecular motors rather than molecular robots.
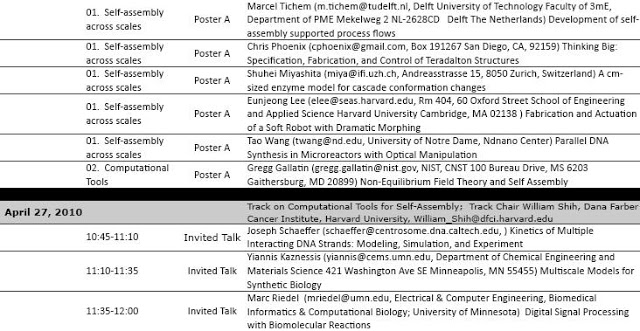
There were a series of talks about self assembly across scales including Chris Phoenix’s own talk.
2. At Metamodern Eric Drexler has a review of Peptoid nanosheets: A platform for new nanotechnologies.
Fresh from Ron Zuckerman’s lab at the Molecular Foundry: a new kind of molecular membrane — thin and crystalline — made by self-assembly of peptoid oligomers. As I discussed in an earlier post, peptoids have remarkable potential as building blocks for self-assembled nanosystems. Peptoids are peptide-like structures, but with monomers that can be chosen from among thousands of readily-available building blocks (a broad class of primary amines).
The design and synthesis of protein-like polymers is a fundamental challenge in materials science. A biomimetic approach is to explore the impact of monomer sequence on non-natural polymer structure and function. We present the aqueous self-assembly of two peptoid polymers into extremely thin two-dimensional (2D) crystalline sheets directed by periodic amphiphilicity, electrostatic recognition and aromatic interactions. Peptoids are sequence-specific, oligo-N-substituted glycine polymers designed to mimic the structure and functionality of proteins. Mixing a 1:1 ratio of two oppositely charged peptoid 36mers of a specific sequence in aqueous solution results in the formation of giant, free-floating sheets with only 2.7 nm thickness. Direct visualization of aligned individual peptoid chains in the sheet structure was achieved using aberration-corrected transmission electron microscopy. Specific binding of a protein to ligand-functionalized sheets was also demonstrated. The synthetic flexibility and biocompatibility of peptoids provide a flexible and robust platform for integrating functionality into defined 2D nanostructures.
36 pages of supplemental information.
If you liked this article, please give it a quick review on Reddit, or StumbleUpon. Thanks
Supporting Advertising
Business Success
How to Make Money
Executive Jobs
Paid Surveys
Thank You

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.