Ad Support : Nano Technology Netbook Technology News Computer Software
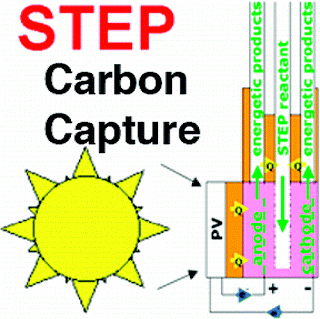
Solar Thermal Electrochemical Photo (STEP) Carbon Capture can be up to 50% efficient at removing CO2 from the atmosphere using solar power.
* 700 square kilometers (270 square miles) of this system would extract the excess CO2 within ten years
* Research is also looking STEP generation of synthetic jet fuel and synthetic diesel
* Further refinement and scaling of STEP carbon capture
* Developing STEP processes to generate a variety of metals and bleach
The first experimental evidence of a new solar process, combining electronic and chemical pathways, to isolate CO2 (carbon capture) is presented. This solar thermal electrochemical photo (STEP) process is a synergy of solid-state and solar thermal processes, and is fundamentally capable of converting more solar energy than photovoltaic or solar thermal processes alone. Here, CO2 is captured using a 750−950 °C electrolysis cell powered by a full spectrum solar simulator in a single step. The process uses the full spectrum; solar thermal energy decreases the energy required for carbon capture, while visible sunlight generates electronic charge to drive the electrolysis. CO2 can be captured from 34% to over 50% solar energy efficiency (depending on the level of solar heat inclusion), as solid carbon and stored, or used as carbon monoxide to be available for a feedstock to synthesize (with STEP generated hydrogen) solar diesel fuel, synthetic jet fuel, or chemical production.
5 pages of supplemental material
From the daily conversion rate of 7.8×10^3 moles of CO2 per square meter of CPV (concentrated photo voltaics), the STEP capture process, scaled to 700 square kilometers of CPV operating for 10 years can remove and convert all this CO2 to carbon. A larger current density at the electrolysis electrodes, will increase the required voltage and would increase the required area of CPVs. Alternatively, a greater degree of solar concentration, for example 2000 suns, rather than 500 suns, will proportionally decrease the area of required CPV area to remove anthropogenic carbon dioxide.
A related resource question is whether there is sufficient lithium carbonate, as an electrolyte of choice for the STEP carbon capture process, to decrease atmospheric levels of carbon dioxide. 700 km2 of CPV plant will generate 5×10^13 Amps of electrolysis current, and require ~2 million metric tonnes of lithium carbonate, as calculated from a 2 kg/l density of lithium carbonate, and assuming that improved, rather than flat, morphology electrodes will operate at 5 A/cm2 (1,000 km2) in a cell of 1 mm thick. Thicker, or lower current density, cells will require proportionally more lithium carbonate. Fifty, rather than ten, years to return the atmosphere to pre-industrial carbon dioxide levels will require proportionally less lithium carbonate. These values are viable within the current production of lithium carbonate. Lithium carbonate availability as a global resource has been under recent scrutiny to meet the growing lithium battery market. It has been estimated that the current global annual production of 0.13 million tonnes of LCE (lithium carbonate equivalents) will increase to 0.24 million tonnes by 2015.SI-1 Potassium carbonate is substantially more available, but as noted in the main portion of the paper can require higher carbon capture electrolysis potentials than lithium carbonate.
If you liked this article, please give it a quick review on Reddit, or StumbleUpon. Thanks
Supporting Advertising
Business Success
How to Make Money
Executive Jobs
Paid Surveys
Thank You

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.