The solar cells are made using nanoparticles — microscopic particles just 30 to 40 atoms across — that are very cheap to produce from zinc and phosphorus, said Jillian Buriak, a University of Alberta chemistry professor and senior research officer of the National Institute of Nanotechnology.
“We turn these things into inks or paints that you can spray coat onto plastics,” Buriak told Quirks & Quarks host Bob McDonald in an interview that airs Saturday.
The efficiency of the solar cells is “not great,” she acknowledged, but that’s something her team is working on.
The fact that they’re “so cheap to make,” she added, means they will only have to reach 7.5 per cent efficiency before they will be commercially competitive with conventional energy sources such as coal-electric generation.
The resulting solar cells can be made extremely light and flexible compared to conventional silicon solar cells.
The zinc phosphide nanoparticle solar cells are also cheaper than conventional solar cells because the process used to make them is very low-energy, Buriak said.
Silicon solar cells are made from sand in a process that involves heating the materials repeatedly to very high temperatures – around 1000 C. As a result, Buriak estimated, it takes three to six years for the resulting solar cell to generate the amount of power used to manufacture it in the first place.
On the other hand, the solar nanoparticles are “actually made in a standard, bubbling pot glassware set up in the lab — the traditional image of chemistry — ” from elements that are very abundant, Buriak said.
ABSTRACT
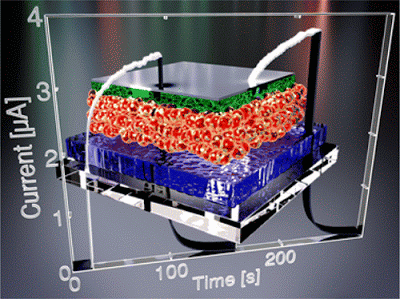
Zinc phosphide (Zn3P2) is a promising earth-abundant material for thin film photovoltaic applications, due to strong optical absorption and near ideal band gap. In this work, crystalline zinc phosphide nanoparticles are synthesized using dimethylzinc and tri-n-octylphosphine as precursors. Transmission electron microscopy and X-ray diffraction data show that these nanoparticles have an average diameter of 8 nm and adopt the crystalline structure of tetragonal α-Zn3P2. The optical band gap is found to increase by 0.5 eV relative to bulk Zn3P2, while there is an asymmetric shift in the conduction and valence band levels. Utilizing layer-by-layer deposition of Zn3P2 nanoparticle films, heterojunction devices consisting of ITO/ZnO/Zn3P2/MoO3/Ag are fabricated and tested for photovoltaic performance. The devices are found to exhibit excellent rectification behavior (rectification ratio of 600) and strong photosensitivity (on/off ratio of 102). X-ray photoelectron spectroscopy and ultraviolet photoemission spectroscopy analyses reveal the presence of a thin 1.5 nm phosphorus shell passivating the surface of the Zn3P2 nanoparticles. This shell is believed to form during the nanoparticle synthesis.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.