Lithium Sulfur Battery could find use in mobile applications, and eventually, electric vehicles with 300-mile range. Researchers at the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) have demonstrated in the laboratory a lithium-sulfur (Li/S) battery that has more than twice the specific energy of lithium-ion batteries, and that lasts for more than 1,500 cycles of charge-discharge with minimal decay of the battery’s capacity. This is longest cycle life reported so far for any lithium-sulfur battery.
Just last month Nextbigfuture reported an advance to 200 charge cycles for lithium sulfur. 1500 charge cycles provide batteries that last about 5-10 years or more. Depending upon the consumers usage of the car and the frequency of charging.
Demand for high-performance batteries for electric and hybrid electric vehicles capable of matching the range and power of the combustion engine encourages scientists to develop new battery chemistries that could deliver more power and energy than lithium-ion batteries, currently the best performing battery chemistry in the marketplace
For electric vehicles to have a 300-mile range, the battery should provide a cell-level specific energy of 350 to 400 Watt-hours/kilogram (Wh/kg). This would require almost double the specific energy (about 200 Wh/kg) of current lithium-ion batteries. The batteries would also need to have at least 1,000, and preferably 1,500 charge-discharge cycles without showing a noticeable power or energy storage capacity loss.
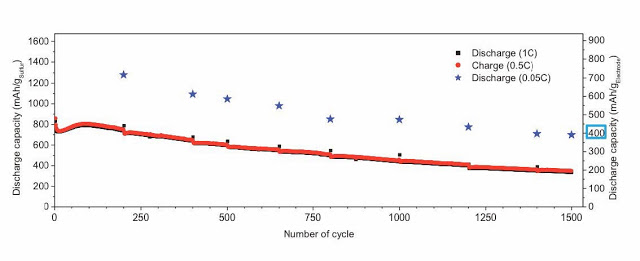
The initial estimated cell-level specific energy of the cell was 500 W·h/kg—much higher than that of current Li-ion cells (~200 W·h/kg). Even after 1,500 cycles, the cell exhibited a very high specific capacity (740 mA·h/g of sulfur), which corresponds to 414 mA·h/g of electrode—still higher than state-of-the-art Li-ion cells. These Li/S cells with lithium metal electrodes can be cycled with an excellent Coulombic efficiency of 96.3% after 1,500 cycles, which, the team said, was enabled by its new formulation of the ionic liquid-based electrolyte.
Lithium-sulfur batteries are attractive for electric vehicles and advanced electronic devices due to their much higher theoretical specific energy (∼2600 W·h/kg) than that of current lithium-ion cells (∼600 W·h/kg). This is due to the very high specific capacity of sulfur (1675 mA·h/g), based on a two-electron reaction (S + 2Li+ + 2e− ↔ Li2S)—significantly larger than the specific capacities of current cathode materials (130−200 mA·h/g).
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.